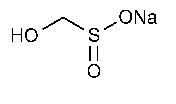
Sodium Formaldehyde Sulfoxylate
Methanesulfinic acid,hydroxy-,monosodium salt.
Monosodium hydroxymethanesulfinate [149-44-0].
Dihydrate 154.11 [6035-47-8].
»Sodium Formaldehyde Sulfoxylate contains an amount of CH3NaO3Sequivalent to not less than 45.5percent and not more than 54.5percent of SO2,calculated on the dried basis.It may contain a suitable stabilizer,such as sodium carbonate.
Packaging and storage—
Preserve in well-closed,light-resistant containers,and store at controlled room temperature.
Clarity and color of solution—
Dissolve 1g in 20mLof water,and transfer 10mLto a 20-×150-mm test tube.Compare with water in a similar test tube:the liquids are equally clear and,when viewed transversely by transmitted light,exhibit no apparent difference in color.
Identification—
A:
Dissolve about 4g in 10mLof water in a test tube,and add 1mLof silver-ammonia-nitrate TS:metallic silver is produced,either as a finely divided,gray precipitate or as a bright metallic mirror on the inner surface of the tube.
B:
Dissolve about 40mg of salicylic acid in 5mLof sulfuric acid,add about 50mg of Sodium Formaldehyde Sulfoxylate,and warm very gently:a permanent,deep red color appears.
Alkalinity—
Dissolve 1.0g in 50mLof water,add phenolphthalein TS,and titrate with 0.10Nsulfuric acid:not more than 3.5mLis required for neutralization.
pHá791ñ:
between 9.5and 10.5,in a solution (1in 50).
Loss on drying á731ñ—
Dry it at 105 for 3hours:it loses not more than 27.0%of its weight.
for 3hours:it loses not more than 27.0%of its weight.
Sulfide—
Dissolve 6g in 14mLof water in a test tube,and wet a strip of lead acetate test paper with the clear solution:no discoloration is evident within 5minutes.
Iron—
Transfer 1.0g to a suitable crucible,and carefully ignite,initially at a low temperature until thoroughly charred,and finally,preferably in a muffle furnace,at 500 to 600
to 600 until the carbon is all burned off.Cool,dissolve the residue in 2mLof hydrochloric acid,and dilute with water to 50mL.Add about 50mg of ammonium persulfate and 5mLof ammonium thiocyanate TS,mix,and transfer to a color-comparison tube.Treat in the same manner 5.0mLof a solution of ferric ammonium sulfate,prepared by dissolving 43.2mg of ferric ammonium sulfate in 10mLof 2Nsulfuric acid and adding water to make 1000mL,each mLrepresenting 5µg of Fe.The color of the test solution is not deeper than that of the solution containing the standard iron solution (0.0025%).
until the carbon is all burned off.Cool,dissolve the residue in 2mLof hydrochloric acid,and dilute with water to 50mL.Add about 50mg of ammonium persulfate and 5mLof ammonium thiocyanate TS,mix,and transfer to a color-comparison tube.Treat in the same manner 5.0mLof a solution of ferric ammonium sulfate,prepared by dissolving 43.2mg of ferric ammonium sulfate in 10mLof 2Nsulfuric acid and adding water to make 1000mL,each mLrepresenting 5µg of Fe.The color of the test solution is not deeper than that of the solution containing the standard iron solution (0.0025%).
Sodium sulfite—
Transfer 4.0mLof the solution prepared for the Assayto a conical flask containing 100mLof water.Add 2mLof formaldehyde TS,and titrate with the same 0.1Niodine VSthat is used for the Assay,adding 3mLof starch TSas the endpoint is approached.Calculate the percentage of Na2SO3in the Sodium Formaldehyde Sulfoxylate taken by the formula:
(1.25)(63.02)(V2-V1)(N/W),
in which 63.02is the equivalent weight of sodium sulfite;V1and V2are the volumes,in mL,of 0.1Niodine VSconsumed in this titration and in the titration performed in the Assay,respectively;Nis the exact normality of the iodine solution;and Wis the weight,in g,of Sodium Formaldehyde Sulfoxylate taken for the Assay:not more than 5.0%of Na2SO3,calculated on the dried basis,is found.
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Assay—
Transfer about 1g of Sodium Formaldehyde Sulfoxylate,accurately weighed,to a 50-mLvolumetric flask,dissolve in about 25mLof water,dilute with water to volume,and mix.Reserve a portion of this solution for the test for Sodium sulfite.Transfer 4.0mLof this solution to a conical flask containing 100mLof water,and titrate with 0.1Niodine VS,adding 3mLof starch TSas the endpoint is approached.Each mLof 0.1Niodine is equivalent to 1.602mg of SO2.
Auxiliary Information—
Staff Liaison:Catherine Sheehan,B.Sc.,Scientist
Expert Committee:(EMC)Excipients:Monograph Content
USP28–NF23Page 3077
Phone Number:1-301-816-8262