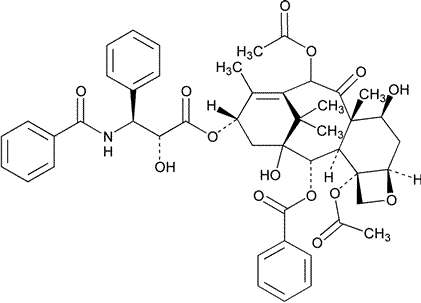
Paclitaxel
C47H51NO14
853.91
Benzenepropanoic acid,b-(benzoylamino)-a-hydroxy-,6,12b-bis(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,11-dihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester,[2aR-[2aa,4b,4ab,6b,9a(aR*,bS*),11a,12a,12aa,12ba]]-.
(2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4,6,9,11,12,12b-hexahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca[3,4]-benz[1,2-b]oxet-5-one 6,12b-diacetate,12-benzoate,9-ester with (2R,3S)-N-benzoyl-3-phenylisoserine [33069-62-4].
Benzenepropanoic acid,b-(benzoylamino)-a-hydroxy-,6,12b-bis(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,11-dihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester,[2aR-[2aa,4b,4ab,6b,9a(aR*,bS*),11a,12a,12aa,12ba]]-.
(2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-1,2a,3,4,4a,6,9,10,11,12,12a,12b-Dodecahydro-4,6,9,11,12,12b-hexahydroxy-4a,8,13,13-tetramethyl-7,11-methano-5H-cyclodeca[3,4]-benz[1,2-b]oxet-5-one 6,12b-diacetate,12-benzoate,9-ester with (2R,3S)-N-benzoyl-3-phenylisoserine [33069-62-4].
»Paclitaxel contains not less than 97.0percent and not more than 102.0percent of C47H51NO14,calculated on the anhydrous,solvent-free basis.
Caution—Paclitaxel is cytotoxic.Great care should be taken to prevent inhaling particles of Paclitaxel and exposing the skin to it.
Packaging and storage—
Preserve in tight,light-resistant containers,and store at controlled room temperature.
Labeling—
The labeling indicates the type of process used to produce the material and theRelated compoundstest with which the material complies.
USP Reference standards á11ñ—
USP Endotoxin RS.USP Paclitaxel RS.USP Paclitaxel Related Compound A RS.USP Paclitaxel Related Compound B RS.
Identification—
A:Infrared Absorption á197Kñ.
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of theStandard preparation,as obtained in theAssay.
Specific rotation á781Sñ:
between -49.0 and -55.0
and -55.0 at 20
at 20 ,calculated on the anhydrous,solvent-free basis.
,calculated on the anhydrous,solvent-free basis.
Test solution:
10mg per mL,in methanol.
Microbial limits á61ñ—
The total aerobic microbial count does not exceed 100cfu per g.It meets the requirements of the tests for the absence ofStaphylococcus aureus,Pseudomonas aeruginosa,Salmonella species,andEscherichia coli.
Bacterial endotoxins á85ñ—
It contains not more than 0.4USP Endotoxin Unit per mg of paclitaxel.
Water,Method Ic á921ñ:
not more than 4.0%.
Residue on ignition á281ñ:
not more than 0.2%.
Heavy metals,Method IIá231ñ:
0.002%.
Related compounds—
TEST1(for material labeled as isolated from natural sources)—
If the material complies with this test,the labeling indicates that it meetsUSP Related compounds Test 1.
Diluent—
Prepare as directed in theAssay.
Solution A—
Prepare filtered and degassed acetonitrile.
Solution B—
Prepare filtered and degassed water.
Mobile phase—
Use variable mixtures ofSolution AandSolution Bas directed forChromatographic system.Make adjustments if necessary (seeSystem Suitability underChromatography á621ñ).
System suitability solution—
Dissolve accurately weighed quantities of USP Paclitaxel Related Compound A RSand USP Paclitaxel Related Compound B RSin methanol to obtain a solution having known concentrations of about 10µg of each per mL.Transfer 5.0mLof this solution to a 50-mLvolumetric flask,dilute withDiluent to volume,and mix.
Standard solution—
Dissolve,with the aid of sonication,an accurately weighed quantity of USP Paclitaxel RSinDiluent,and dilute quantitatively,and stepwise if necessary,withDiluent to obtain a solution having a known concentration of about 5µg per mL.
Test solution—
Use theAssay preparation.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 227-nm detector and a 4.6-mm ×25-cm column that contains 5-µm packing L43.The flow rate is about 2.6mLper minute.The column temperature is maintained at 30 .The chromatograph is programmed as follows.
.The chromatograph is programmed as follows.
Chromatograph theSystem suitability solution,and record the peak responses as directed forProcedure:the relative retention times are about 0.78for paclitaxel related compound Aand 0.86for paclitaxel related compound B(relative to the retention time for paclitaxel obtained from theTest solution);and the resolution,R,between paclitaxel related compound Aand paclitaxel related compound Bis not less than 1.0.Chromatograph theStandard solution,and record the peak responses as directed forProcedure:the relative standard deviation for replicate injections is not more than 2.0%.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
| 0–35 | 35 | 65 | isocratic |
| 35–60 | 35®80 | 65®20 | linear gradient |
| 60–70 | 80®35 | 20®65 | linear gradient |
| 70–80 | 35 | 65 | isocratic |
Procedure—
Inject a volume (about 15µL)of theTest solution into the chromatograph,record the chromatogram,and measure the areas for the major peaks.Calculate the percentage of each impurity in the portion of Paclitaxel taken by the formula:
In addition to not exceeding the limits for paclitaxel related impurities inTable 1,not more than 0.1%of any other single impurity is found;and not more than 2.0%of total impurities is found.
100(Fri/rU),
in whichFis the relative response factor for each impurity peak (seeTable 1for values);riis the peak area for each individual impurity;andrUis the peak area for paclitaxel.
Table 1
| Relative Retention Time |
Relative Response Factor (F) |
Name | Limit (%) |
| 0.24 | 1.29 | Baccatin III | 0.2 |
| 0.53 | 1.00 | 10-Deacetylpaclitaxel | 0.5 |
| 0.57 | 1.00 | 7-Xylosylpaclitaxel | 0.2 |
| 0.78 | 1.26 | Cephalomannine (paclitaxel related compound A) | a11 |
| 0.78 | 1.26 | 2¢¢,3¢¢-Dihydrocephalomannine | a21 |
| 0.86 | 1.00 | 10-Deacetyl-7-epipaclitaxel (paclitaxel related compound B) | 0.5 |
| 1.10 | 1.00 | Benzyl analog3 | b12 |
| 1.10 | 1.00 | 3¢¢,4¢¢-Dehydropaclitaxel C | b22 |
| 1.40 | 1.00 | 7-Epicephalomannine | 0.3 |
| 1.85 | 1.00 | 7-Epipaclitaxel | 0.5 |
|
1
Resolution may be incomplete for these peaks,depending upon the relative amounts present;the sum of a1and a2is not more than 0.5%.
2
Resolution may be incomplete for these peaks depending upon the relative amounts present;the sum of b1and b2is not more than 0.5%.
3
The following chemical name is assigned to the related compound,benzyl analog:Baccatin III13-ester with (2R,3S)-2-hydroxy-3-phenyl-3-(2-phenylacetylamino)propanoic acid.
|
|||
TEST2(for material labeled as produced by a semisynthetic process)—
If the material complies with this test,the labeling indicates that it meetsUSP Related compounds Test 2.
Diluent—
Use acetonitrile.
Solution A—
Use a filtered and degassed mixture of water and acetonitrile (3:2).
Solution B—
Use filtered and degassed acetonitrile.
Mobile phase—
Use variable mixtures ofSolution AandSolution Bas directed forChromatographic system.Make adjustments if necessary (seeSystem Suitability underChromatography á621ñ).
System suitability solution—
Dissolve accurately weighed quantities of USP Paclitaxel RSand USP Paclitaxel Related Compound B RSinDiluent,using shaking and sonication if necessary,to obtain a solution having known concentrations of about 0.96mg and 0.008mg per mL,respectively.
Test solution—
Transfer about 10mg of Paclitaxel,accurately weighed,to a 10-mLvolumetric flask;dissolve in and dilute with Diluentto volume,using shaking and sonication if necessary;and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 227-nm detector and a 4.6-mm ×15-cm column that contains 3-µm packing L1.The flow rate is about 1.2mLper minute.The column temperature is maintained at 35 .The chromatograph is programmed as follows.
.The chromatograph is programmed as follows.
Chromatograph theSystem suitability solution,and record the peak responses as directed forProcedure:the relative retention times are about 0.94for paclitaxel related compound Band 1.0for paclitaxel;the resolution,R,between paclitaxel related compound Band paclitaxel is not less than 1.2;and the relative standard deviation for replicate injections is not more than 2.0%.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
| 0–20 | 100 | 0 | isocratic |
| 20–60 | 100®10 | 0®90 | linear gradient |
| 60–62 | 10®100 | 90®0 | linear gradient |
| 62–70 | 100 | 0 | isocratic |
Procedure—
Separately inject equal volumes (about 15µL)of theDiluent and theTest solution into the chromatograph,record the chromatograms,and measure the areas for all the peaks.Disregard any peaks due to theDiluent.Calculate the percentage of each impurity in the portion of Paclitaxel taken by the formula:
In addition to not exceeding the limits for paclitaxel related impurities in Table 2,not more than 0.1%of any other single impurity is found;and not more than 2.0%of total impurities is found.
100(Fri/rs),
in whichFis the relative response factor for each impurity (seeTable 2for values);riis the peak area for each impurity obtained from theTest solution;andrsis the sum of the areas of all the peaks obtained from theTest solution.
Table 2
| Relative Retention Time |
Relative Response factor (F) |
Name | Limit (%) |
| 0.11 | 1.24 | 10-Deacetylbaccatin III | 0.1 |
| 0.20 | 1.29 | Baccatin III | 0.2 |
| 0.42 | 1.39 | Photodegradant2 | 0.1 |
| 0.47 | 1.00 | 10-Deacetylpaclitaxel | 0.5 |
| 0.80 | 1.00 | 2-Debenzoylpaclitaxel-2-pentenoate | 0.7 |
| 0.921 | 1.00 | Oxetane ring opened,acetyl and benzoyl2 | x1 |
| 0.921 | 1.00 | 10-Acetoacetylpaclitaxel | x2 |
| 0.941 | 1.00 | 10-Deacetyl-7-epipaclitaxel (paclitaxel related compound B) | x3 |
| 1.37 | 1.00 | 7-Epipaclitaxel | 0.4 |
| 1.45 | 1.00 | 10,13-Bissidechainpaclitaxel2 | 0.5 |
| 1.54 | 1.00 | 7-Acetylpaclitaxel | 0.6 |
| 1.80 | 1.75 | 13-Tes-baccatin III | 0.1 |
| 2.14 | 1.00 | 7-Tes-paclitaxel | 0.3 |
|
1
Resolution may be incomplete for these peaks depending upon the relative amounts present;the sum of x1,x2,and x3is not more than 0.4%.
2
The following chemical names are assigned to the related compounds Photodegradant,Oxetane ring opened,acetyl and benzoyl,and 10,13-Bissidechainpaclitaxel:
Photodegradant
(1R,2R,4S,5S,7R,10S,11R,12S,13S,15S,16S)-2,10-diacetyloxy-5,13-dihydroxy-4,16,17,17-tetramethyl-8-oxa-3-oxo-12-phenylcarbonyloxypentacyclo[11.3.1.01,11.04,11.07,10]heptadec-15-yl
(2R,3S)-2-hydroxy-3-phenyl-3-(phenylcarbonylamino)propanoate
Oxetane ring opened,acetyl and benzoyl migrated
(1S,2S,3R,4S,5S,7S,8S,10R,13S)-5,10-diacetyloxy-1,2,4,7-tetrahydroxy-8,12,15,15-tetramethyl-9-oxo-4-(phenylcarbonyloxymethyl)tricyclo[9.3.1.03,8]pentadec-11-en-13-yl
(2R,3S)-2-hydroxy-3-phenyl-3-(phenylcarbonylamino)propanoate
10,13-Bissidechainpaclitaxel
Baccatin III13-ester with (2R,3S)-2-hydroxy-3-phenyl-3-(phenylcarbonylamino)propanoic acid,10-ester with (2S,3S)-2-hydroxy-3-phenyl-3-(phenylcarbonylamino)propanoic acid
|
|||
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Diluent—
Prepare a mixture of methanol and acetic acid (200:1).
Mobile phase—
Prepare a filtered and degassed mixture of water and acetonitrile (11:9).Make adjustments if necessary (seeSystem Suitability underChromatography á621ñ).
Standard preparation—
Dissolve,using sonication if necessary,an accurately weighed quantity of USP Paclitaxel RSinDiluent,and dilute quantitatively,and stepwise if necessary,withDiluent to obtain a solution having a known concentration of about 1mg per mL.
Assay preparation—
Transfer about 10mg of Paclitaxel,accurately weighed,to a 10-mLvolumetric flask.Dissolve inDiluent,using sonication if necessary,dilute withDiluent to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 227-nm detector and a 4.6-mm ×25-cm column that contains 5-µm packing L43.The flow rate is about 1.5mLper minute.Chromatograph theStandard preparation,and record the peak responses as directed forProcedure:the tailing factor is between 0.7and 1.3;and the relative standard deviation for replicate injections is not more than 1.5%.
Procedure—
Separately inject equal volumes (about 10µL)of theStandard preparation and theAssay preparation into the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the quantity,in mg,of C47H51NO14in the portion of Paclitaxel taken by the formula:
10C(rU/rS),
in whichCis the concentration,in mg per mL,of USP Paclitaxel RSin theStandard preparation;andrUandrSare the peak responses for paclitaxel obtained from theAssay preparation and theStandard preparation,respectively.
Auxiliary Information—
Staff Liaison:Lawrence Evans,III,Ph.D.,Scientist
Expert Committee:(PA6)Pharmaceutical Analysis 6
USP28–NF23Page 1456
Pharmacopeial Forum:Volume No.30(4)Page 1279
Phone Number:1-301-816-8389