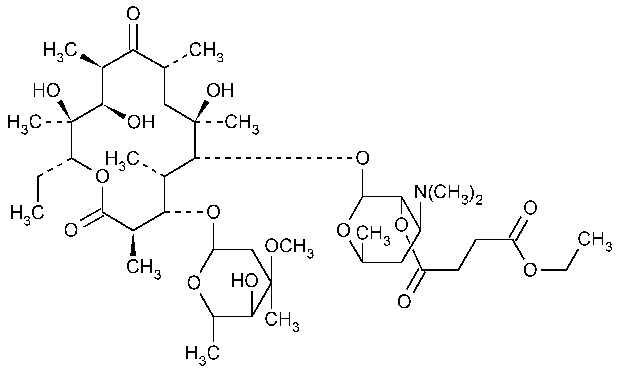
Erythromycin Ethylsuccinate
Erythromycin 2¢-(ethyl butanedioate).
Erythromycin 2¢-(ethyl succinate). [1264-62-6].
»Erythromycin Ethylsuccinate consists primarily of the 2¢-ethylsuccinate ester of erythromycin A.The sum of the percentages of erythromycin A,erythromycin B,and erythromycin Cis not less than 76.5percent,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
Labeling—
Erythromycin Ethylsuccinate that is noncrystalline is labeled to indicate that it is amorphous.Any preparation containing the amorphous form of Erythromycin Ethylsuccinate is so labeled.
USP Reference standards á11ñ—
USP Erythromycin RS.USP Erythromycin B RS.USP Erythromycin C RS.USP Erythromycin Related Compound N RS.USP Erythromycin Ethylsuccinate RS.
Identification,Infrared Absorptioná197Sñ—
Solution:
1in 100.
Medium:
chloroform.
Cell size:
1.0-mm.
Crystallinity á695ñ:
meets the requirements,except that when it is labeled as being in the amorphous state it does not meet the requirements.
X-ray diffraction á941ñ—
Where labeled as being in the amorphous state,its X-ray diffraction pattern performed at high sensitivity for angles of diffraction between 2 and 20
and 20 exhibits no reflection,and between 7
exhibits no reflection,and between 7 and 10
and 10 exhibits a more intense hachured baseline,creating a halo.
exhibits a more intense hachured baseline,creating a halo.
Water,Method Iá921ñ:
not more than 3.0%,20mLof methanol containing 10%of imidazole being used in place of methanol in the titration vessel.
Residue on ignition á281ñ:
not more than 1.0%after ignition at 550±50 ,the charred residue being moistened with 2mLof nitric acid and 5drops of sulfuric acid.
,the charred residue being moistened with 2mLof nitric acid and 5drops of sulfuric acid.
Related compounds—
Using the chromatograms obtained for the Assay preparationand Standard preparation 2in the Assay,begin peak integration after the two peaks for succinates that elute just after the solvent front,and calculate the percentage of each related compound having the greatest response,other than erythromycin A,erythromycin B,erythromycin C,erythromycin Aenol ether,and erythromycin N-ethylsuccinate (retention time relative to erythromycin Apeak is about 1.3),in the portion of Erythromycin Ethylsuccinate taken by the formula:
50(CS2P/W)(ri/rS2),
in which CS2is the concentration,in mg per mL,of USP Erythromycin RSin Standard preparation 2;Pis the designated percentage of erythromycin Ain USP Erythromycin RS;Wis the quantity,in mg,of Erythromycin Ethylsuccinate taken to prepare the Assay preparation;riis the peak response of each related compound,other than erythromycin A,erythromycin B,erythromycin C,erythromycin Aenol ether,and erythromycin N-ethylsuccinate,in the chromatogram obtained from the Assay preparation;and rS2is the erythromycin Apeak response in the chromatogram obtained from Standard preparation 2:not more than 3.0%of any individual related compound is found.
Calculate the percentage of erythromycin Aenol ether in the portion of Erythromycin Ethylsuccinate taken by the formula:
(50/11)(CS2P/W)(rE/rS2),
in which 11is the response factor for erythromycin Aenol ether in relation to that of erythromycin A;rEis the peak response of the erythromycin Aenol ether observed in the chromatogram obtained from the Assay preparation;and the other terms are as defined above:not more than 3.0%of erythromycin Aenol ether is found.
Calculate the percentage of erythromycin N-ethylsuccinate in the portion of Erythromycin Ethylsuccinate taken by the formula:
(50/7.4)(CS2P/W)(rN/rS2),
in which 7.4is the response factor for erythromycin N-ethylsuccinate in relation to that of erythromycin A;rNis the peak response of the erythromycin N-ethylsuccinate (retention time relative to the erythromycin Apeak is about 1.3)observed in the chromatogram obtained from the Assay preparation;and the other terms are as defined above:not more than 3.0%of erythromycin N-ethylsuccinate is found.
Assay—
Hydrolysis reagent—
Prepare a solution of dibasic potassium phosphate (2in 100),and adjust with phosphoric acid to a pHof 8.0.
pH8.0Buffer—
Prepare a solution of dibasic potassium phosphate (3.5in 100),and adjust with phosphoric acid to a pHof 8.0.
pH3.5Buffer—
Adjust 20mLof pH8.0Bufferwith phosphoric acid to a pHof 3.5.
Mobile phase—
Mix 50mLof pH8.0Bufferwith 400mLof water,add 175mLof tertiary butyl alcohol and 30mLof acetonitrile,dilute with water to 1000mL,and mix.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
NOTE—Use the following solutions promptly,or within 1day if stored in a refrigerator.
Standard preparation 1—
Transfer about 50mg of USP Erythromycin RS,accurately weighed,to a 25-mLvolumetric flask,add 12.5mLof methanol,and swirl to dissolve.Dilute with Hydrolysis reagentto volume,and mix.
Standard preparation 2—
Transfer about 5mg each of USP Erythromycin B RSand USP Erythromycin C RS,both accurately weighed,to a 50-mLvolumetric flask,add 25.0mLof methanol,and swirl to dissolve.Add 2.5mLof Standard preparation 1,dilute with Hydrolysis reagentto volume,and mix.
System suitability solution—
Dissolve about 2mg of USP Erythromycin Related Compound N RSin about 20mLof Standard preparation 2,and mix.
Erythromycin Aenol ether solution—
Dissolve about 10mg of USP Erythromycin RSin 2mLof methanol.Add 10mLof pH3.5Buffer,mix,and allow to stand for about 30minutes.Refrigerate this solution until used,and discard 8hours after preparation.
Assay preparation—
Transfer about 115mg of Erythromycin Ethylsuccinate,accurately weighed,to a 50-mLvolumetric flask,add 25.0mLof methanol,and swirl to dissolve.Add about 20mLof Hydrolysis reagent,mix,and allow to stand at room temperature for about 12hours to effect hydrolysis.Dilute with Hydrolysis reagentto volume,and mix.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 215-nm detector and a 4.6-mm ×25-cm column that contains packing L21(1000Å)and is maintained at a constant temperature of about 70 .The flow rate is about 2mLper minute.Chromatograph the System suitability solution,and record the peak responses as directed for Procedure:the order of elution of the components is erythromycin related compound N,erythromycin C,erythromycin A,and erythromycin B;and the resolution,R,between erythromycin related compound Nand erythromycin Cis not less than 0.8and between erythromycin related compound Nand erythromycin Anot less than 5.5.Chromatograph the Erythromycin Aenol ether solution,and record the peak responses as directed for Procedure:adjust the duration of chromatography to include the erythromycin Aenol ether peak,which has a retention time of about 4.3to 4.7times that of erythromycin A.Chromatograph Standard preparation 1,and record the peak responses as directed for Procedure:the relative standard deviation for replicate injections is not more than 1.0%.
.The flow rate is about 2mLper minute.Chromatograph the System suitability solution,and record the peak responses as directed for Procedure:the order of elution of the components is erythromycin related compound N,erythromycin C,erythromycin A,and erythromycin B;and the resolution,R,between erythromycin related compound Nand erythromycin Cis not less than 0.8and between erythromycin related compound Nand erythromycin Anot less than 5.5.Chromatograph the Erythromycin Aenol ether solution,and record the peak responses as directed for Procedure:adjust the duration of chromatography to include the erythromycin Aenol ether peak,which has a retention time of about 4.3to 4.7times that of erythromycin A.Chromatograph Standard preparation 1,and record the peak responses as directed for Procedure:the relative standard deviation for replicate injections is not more than 1.0%.
Procedure—
Separately inject equal volumes (about 200µL)of Standard preparation 1,Standard preparation 2,and the Assay preparationinto the chromatograph;record the chromatograms for a period of time that is adequate to include the erythromycin Aenol ether peak,if present;and measure the areas of the peak responses.Calculate the percentage of erythromycin Ain the portion of Erythromycin Ethylsuccinate taken by the formula:
50(CS1P/W)(rU/rS1),
in which CS1is the concentration,in mg per mL,of USP Erythromycin RSin Standard preparation 1;Pis the designated percentage of erythromycin Ain USP Erythromycin RS;Wis the quantity,in mg,of Erythromycin Ethylsuccinate taken to prepare the Assay preparation;and rUand rS1are the erythromycin Apeak responses in the chromatograms obtained from the Assay preparationand Standard preparation 1,respectively.
Calculate the percentage of erythromycin Band erythromycin Cin the portion of Erythromycin Ethylsuccinate taken by the formula:
50(CS2P/W)(rU/rS2),
in which CS2is the concentration,in mg per mL,of the relevant USP Reference Standard in Standard preparation 2;Pis the designated percentage of erythromycin Bor erythromycin Cin the relevant USP Reference Standard;Wis the quantity,in mg,of Erythromycin Ethylsuccinate taken to prepare the Assay preparation;and rUand rS2are the peak responses of the relevant analyte in the chromatograms obtained from the Assay preparationand Standard preparation 2,respectively.The percentage of erythromycin Bis not more than 12.0%,and the percentage of erythromycin Cis not more than 5.0%.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 765
Phone Number:1-301-816-8335