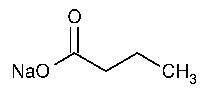
Sodium Butyrate
»Sodium Butyrate contains not less than 98.0percent and not more than 101.0percent of C4H7NaO2,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers,and store at controlled room temperature.
Labeling—
Label it to indicate that it is intended for use in compounding dosage forms for rectal use only.
Identification—
A:
Infrared Absorption á197Kñ,on the undried specimen.
B:
Asolution (1in 10)meets the requirements of the tests for Sodium á191ñ.
Alkalinity—
Dissolve 2.0g in 20mLof water,and add 1drop of phenolphthalein TS:if a pink color is produced,it is discharged by 0.50mLof 0.10Nsulfuric acid.
Water,Method Iá921ñ:
not more than 1.0%.
Heavy metals,Method IIá231ñ:
0.001%.
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Assay—
Dissolve about 200mg of Sodium Butyrate,accurately weighed,in 50mLof glacial acetic acid.Add 1drop of crystal violet TS,and titrate with 0.1Nperchloric acid to a green endpoint.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 11.01mg of C4H7NaO2.
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(PA4)Pharmaceutical Analysis 4
USP28–NF23Page 1778
Pharmacopeial Forum:Volume No.28(6)Page 1868
Phone Number:1-301-816-8251