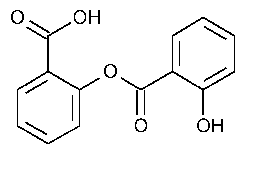
Salsalate
Benzoic acid,2-hydroxy-,2-carboxyphenyl ester.
Disalicylic acid.
Salicylsalicylic acid.
Salicylic acid,bimolecular ester [552-94-3].
»Salsalate contains not less than 98.0percent and not more than 102.0percent of total salicylates,expressed as the sum of the percentages of salsalate,salicylic acid,and trisalicylic acid,calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification,Infrared Absorption á197Mñ.
Loss on drying á731ñ—
Dry it in vacuum at 60 for 3hours:it loses not more than 0.5%of its weight.
for 3hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.10%.
Chloride á221ñ—
Dissolve 1.4g in 6mLof methanol,warming if necessary to effect solution.Dilute with water to 50mLto precipitate the salsalate,allow to stand for 5minutes,and filter.A25-mLportion of the filtrate shows no more chloride than corresponds to 0.20mLof 0.010Nhydrochloric acid (0.01%).
Sulfate á221ñ—
A17-mLportion of the filtrate prepared for the test for Chlorideshows no more sulfate than corresponds to 0.50mLof 0.010Nsulfuric acid (0.05%).
Heavy metals—
Dissolve 2g in 25mLof methanol,and add 1mLof water.Add 1.2mLof thioacetamide-glycerin base TSand 2mLof pH3.5Acetate Buffer,and allow to stand for 5minutes:any color produced is not darker than that obtained from a standard prepared from 25mLof methanol and 2mLof Standard Lead Solution(see Heavy metals á231ñ)and treated in the same manner.The limit is 10µg per g.
Limit of dimethylaniline—
Internal standard solution—
Prepare a solution in methylene chloride containing 0.4mg of indene per mL.
Standard preparation—
Transfer about 50mg of N,N-dimethylaniline,accurately weighed,to a 100-mLvolumetric flask,dilute with Internal standard solutionto volume,insert the stopper securely,and mix.
Test preparation—
Transfer about 5g of Salsalate,accurately weighed,to a 125-mLseparator fitted with a cotton pledget in its stem.Add 50mLof water and 6mLof 6Nammonium hydroxide,and swirl until dissolved.Add 5.0mLof Internal standard solution,insert the stopper into the separator,and shake for 1minute.Keep the separator stoppered,and allow the layers to separate.Loosen the stopper,and drain most of the lower phase into a screw-capped test tube.Use this solution as the Test preparation.
Chromatographic system
(see Chromatography á621ñ)—The gas chromatograph is equipped with a flame-ionization detector,a split injector with a 10:1split ratio,and a 30-m ×0.53-mm capillary column,the internal wall of which is coated with a 1.0-µm film of phase G42.Maintain the column at 105 ,the injector at 250
,the injector at 250 ,and the detector block at 250
,and the detector block at 250 ,and use helium as the carrier gas,at a flow rate of about 13mLper minute.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the relative retention times are about 0.75for indene and 1.0for N,N-dimethylaniline,the resolution,R,between the indene peak and the N,N-dimethylaniline peak is not less than 2.0,and the relative standard deviation for replicate injections is not more than 3%.
,and use helium as the carrier gas,at a flow rate of about 13mLper minute.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the relative retention times are about 0.75for indene and 1.0for N,N-dimethylaniline,the resolution,R,between the indene peak and the N,N-dimethylaniline peak is not less than 2.0,and the relative standard deviation for replicate injections is not more than 3%.
Procedure—
[NOTE—Use peak areas where peak responses are indicated.]Inject equal volumes (about 1µL)of the Standard preparationand the Test preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Indene elutes before N,N-dimethylaniline.Calculate the percentage of N,N-dimethylaniline in the portion of Salsalate taken by the formula:
0.5(C/W)(RU/RS),
in which Cis the concentration,in mg per mL,of N,N-dimethylaniline in the Standard preparation,Wis the weight,in g,of Salsalate taken to prepare the Test preparation,and RUand RSare the ratios of the response of the N,N-dimethylaniline peak to that of the indene peak obtained from the Test preparationand the Standard preparation,respectively.The limit is 0.05%.
Isopropyl,ethyl,and methyl salicylates—
Standard stock solution—
Prepare a solution in chromatographic n-heptane containing 0.20mg of isopropyl salicylate,0.50mg of ethyl salicylate,and 0.50mg of methyl salicylate per mL.
Standard preparation—
Transfer to a suitable screw-capped test tube 2.0g of Salsalate,add 10mLof 1Nsodium hydroxide and 2mLof chromatographic n-heptane,shake until dissolved,and allow the layers to separate.Draw off and discard all of the upper layer.To the lower layer add 2.0mLof Standard stock solution,shake for 1minute,and allow the layers to separate.Use the upper layer as the Standard preparation.
Test preparation—
Transfer 2.0g of Salsalate to a suitable screw-capped test tube,add 10mLof 1Nsodium hydroxide and 2.0mLof chromatographic n-heptane,shake until dissolved,and allow the layers to separate.Use the upper layer as the Test preparation.
Chromatographic system
(see Chromatography á621ñ)—The gas chromatograph is equipped with a flame-ionization detector,a split injector with a 10:1split ratio,and a 30-m ×0.53-mm capillary column,the internal wall of which is coated with a 1.0-µm film of phase G42.Maintain the column at 120 and the injector and detector block at about 250
and the injector and detector block at about 250 .Helium is used as the carrier gas,flowing at the rate of about 13mLper minute.
.Helium is used as the carrier gas,flowing at the rate of about 13mLper minute.
Procedure—
[NOTE—Use peak areas where peak responses are indicated.]Inject equal volumes (about 1µL)of the Standard preparationand the Test preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.The relative retention times are about 0.65for methyl salicylate,0.9for ethyl salicylate,and 1.0for isopropyl salicylate.The response of any isopropyl salicylate peak obtained from the Test preparationis not greater than that obtained from the Standard preparation(0.02%),the response of any ethyl salicylate peak obtained from the Test preparationis not greater than that obtained from the Standard preparation(0.05%),and the response of any methyl salicylate peak obtained from the Test preparationis not greater than that obtained from the Standard preparation(0.05%).
Chromatographic purity—
Using the chromatograms obtained in the Assay,calculate the percentage of each impurity,other than salicylic acid and trisalicylic acid,in the portion of Salsalate taken by the formula:
10,000(C/W)(rU/rS),
in which Cis the concentration,in mg per mL,of USP Salsalate RSin the Salsalate standard preparation,Wis the weight,in mg,of Salsalate taken to prepare the Assay stock solution,rUis the response of the particular impurity peak obtained from the Assay stock solution,and rSis the salsalate peak response obtained from the Salsalate standard preparation:not more than 0.2%of each other impurity is found.
Related compounds—
The percentages of salicylic acid and trisalicylic acid,determined as directed in the Assay,do not exceed 0.5%and 2.5%,respectively.
Organic volatile impurities,Method Vá467ñ:
meets the requirements.
Solvent—
Use dimethyl sulfoxide.
Assay—
Mobile phase—
Prepare a suitable filtered and degassed mixture of methanol,water,and phosphoric acid (650:350:1),and adjust with phosphoric acid or 1Nsodium hydroxide,if necessary,to a pHof 3.1.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Diluent—
Prepare a mixture of water,acetonitrile,and phosphoric acid (540:460:1).
Salsalate standard preparation—
Dissolve an accurately weighed quantity of USP Salsalate RSin Diluentto obtain a stock solution having a known concentration of about 1mg per mL.Transfer 2.0mLof this solution to a 100-mLvolumetric flask,dilute with Diluentto volume,and mix.This solution contains about 0.02mg per mL.
Salicylic acid standard preparation—
Dissolve an accurately weighed quantity of USP Salicylic Acid RSin Diluentto obtain a stock solution having a known concentration of about 0.5mg per mL.Transfer 1.0mLof this solution to a 100-mLvolumetric flask,dilute with Diluentto volume,and mix.This solution contains about 0.005mg of USP Salicylic Acid RSper mL.
Trisalicylic acid standard preparation—
Dissolve an accurately weighed quantity of USP Trisalicylic Acid RSin Diluentto obtain a stock solution having a known concentration of about 0.5mg per mL.Transfer 5.0mLof this solution to a 100-mLvolumetric flask,dilute with Diluentto volume,and mix.This solution contains about 0.025mg of USP Trisalicylic Acid RSper mL.
Resolution solution—
Prepare a solution in Diluentcontaining about 0.02mg of USP Salsalate RSper mL,0.02mg of USP Salicylic Acid RSper mL,and 0.04mg of USP Trisalicylic Acid RSper mL.
Assay stock solution—
Transfer about 100mg of Salsalate,accurately weighed,to a 100-mLvolumetric flask,dilute with Diluentto volume,and mix.Sonicate if necessary to effect the solution.
Assay preparation—
Transfer 2.0mLof the Assay stock solutionto a 100-mLvolumetric flask,dilute with Diluentto volume,and mix.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 236-nm detector and a 4-mm ×15-cm column that contains 5-µm packing L7.The flow rate is about 1.5mLper minute.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 0.55for salicylic acid,1.0for salsalate,and 1.5for trisalicylic acid,and the resolution,R,between the salicylic acid and salsalate peaks and between the salsalate and trisalicylic acid peaks is not less than 2.0.Chromatograph the Salicylic acid standard preparation,and record the peak responses as directed for Procedure:the relative standard deviation of the salicylic acid peak responses for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10µL)of the Salsalate standard preparation,the Salicylic acid standard preparation,the Trisalicylic acid standard preparation,the Assay stock solution,and the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.[NOTE—Continue chromatography after each injection for a period of time not less than the retention time of trisalicylic acid.]Calculate the percentage of salicylic acid (C7H6O3)in the portion of Salsalate taken by the formula:
10,000(C/W)(rU/rS),
in which Cis the concentration,in mg per mL,of USP Salicylic Acid RSin the Salicylic acid standard preparation,Wis the weight,in mg,of the portion of Salsalate taken,and rUand rSare the responses of the salicylic acid peak obtained from the Assay stock solutionand the Salicylic acid standard preparation,respectively.Calculate the percentage of trisalicylic acid (C21H14O7)in the portion of Salsalate taken by the formula:
10,000(C/W)(rU/rS),
in which Cis the concentration,in mg per mL,of USP Trisalicylic Acid RSin the Trisalicylic acid standard preparation,and rUand rSare the responses of the trisalicylic acid peaks obtained from the Assay stock solutionand the Trisalicylic acid standard preparation,respectively.Calculate the percentage of salsalate (C14H10O5)in the portion of Salsalate taken by the formula:
500,000(C/W)(rU/rS),
in which Cis the concentration,in mg per mL,of USP Salsalate RSin the Salsalate standard preparation,and rUand rSare the salsalate peak responses obtained from the Assay preparationand the Salsalate standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Daniel K.Bempong,Ph.D.,Scientist
Expert Committee:(PA2)Pharmaceutical Analysis 2
USP28–NF23Page 1749
Phone Number:1-301-816-8143