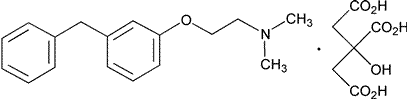
Add the following:
»Phenyltoloxamine Citrate contains not less than 99.0percent and not more than 101.0percent of C17H21NO·C6H8O7,calculated on the dried basis.
USP Reference standards á11ñ—
USP Phenyltoloxamine Citrate RS.USP Phenyltoloxamine Related Compound A RS.
Identification,
Infrared Absorption á197Kñ.
pHá791ñ:
between 3.2and 4.2,in a solution (1in 100).
Loss on drying á731ñ—
Dry it in vacuum at 80 for 3hours:it loses not more than 0.5%of its weight.
for 3hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Heavy metals,Method Iá231ñ:
20µg per g.
Related compounds—
Resolution solution—
In a separatory funnel dissolve about 10mg each of USP Phenyltoloxamine Citrate RSand USP Phenyltoloxamine Related Compound A RS,accurately weighed,in 50mLof water.Add 5mLof ammonium hydroxide,and extract with three 10-mLportions of methylene chloride.Combine the extracts,dry the solution over anhydrous sodium sulfate,and gently evaporate to dryness.Dissolve the residue in 20mLof methylene chloride.
Test solution—
In a separatory funnel dissolve about 400mg of Phenyltoloxamine Citrate,accurately weighed,in 50mLof water.Proceed as directed for Resolution solution,beginning with “Add 5mLof ammonium hydroxide.”
Chromatographic system (see Chromatography á621ñ)—
The gas chromatograph is equipped with a split injection system,a flame-ionization detector,and a 0.32-mm ×25-m column coated with a 0.45-µm film of phase G27.The carrier gas is helium,flowing at a rate of about 29cm per second,with a split flow rate of about 25mLper minute.The column temperature is programmed as follows.Initially the temperature of the column is equilibrated at 190 for 3minutes,then the temperature is increased at a rate of 4
for 3minutes,then the temperature is increased at a rate of 4 per minute to 240
per minute to 240 ,and maintained at 240
,and maintained at 240 for 8minutes.The injection port and the detector temperatures are maintained at 280
for 8minutes.The injection port and the detector temperatures are maintained at 280 .Chromatograph theResolution solution,and record the peak responses as directed for Procedure:the resolution,R,between phenyltoloxamine and phenyltoloxamine related compound Ais not less than 1.5.
.Chromatograph theResolution solution,and record the peak responses as directed for Procedure:the resolution,R,between phenyltoloxamine and phenyltoloxamine related compound Ais not less than 1.5.
Procedure—
Inject a volume (about 1µL)of the Test solutioninto the chromatograph,record the chromatograms,and measure the peak responses.Calculate the percentage of each impurity in the portion of Phenyltoloxamine Citrate taken by the formula:
100(ri/rs),
in which riis the peak response of each impurity;and rsis the sum of the responses of all the peaks,excluding the solvent peaks:not more than 0.2%of phenyltoloxamine related compound A;not more than 0.1%of any other individual impurity;and not more than 1.0%of total impurities is found.
Organic volatile impurities,Method 1á467ñ:
meets the requirements.
Assay—
Dissolve about 0.5g of Phenyltoloxamine Citrate,accurately weighed,in 80mLof glacial acetic acid,and titrate with 0.1Nperchloric acid VS,determining the endpoint potentiometrically.Perform a blank determination,and make any necessary correction (see Titrimetry á541ñ).Each mLof 0.1Nperchloric acid is equivalent to 44.75mg of C17H21NO·C6H8O7. USP28
USP28
Auxiliary Information—
Staff Liaison:Karen A Russo,Ph.D.,Scientist
Expert Committee:(PA1)Pharmaceutical Analysis 1
USP28–NF23Page 1547
Pharmacopeial Forum:Volume No.30(4)Page 1291
Phone Number:1-301-816-8379