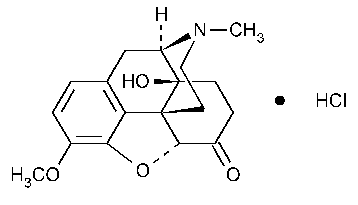
Oxycodone Hydrochloride
Morphinan-6-one,4,5-epoxy-14-hydroxy-3-methoxy-17-methyl-,hydrochloride,(5a)-.
4,5a-Epoxy-14-hydroxy-3-methoxy-17-methylmorphinan-6-one hydrochloride [124-90-3].
»Oxycodone Hydrochloride contains not less than 97.0percent and not more than 103.0percent of C18H21NO4·HCl,calculated on the anhydrous,solvent-free basis.
Packaging and storage—
Preserve in tight containers.
Identification—
A:
Dissolve 250mg in 25mLof water.Render the solution alkaline with 6Nammonium hydroxide.Allow the mixture to stand until a precipitate is formed.Filter,wash the precipitate with 50mLof cold water,and dry for 2hours at 105 :the precipitate so obtained melts between 218
:the precipitate so obtained melts between 218 and 223
and 223 ,but the range between beginning and end of melting does not exceed 2
,but the range between beginning and end of melting does not exceed 2 (see Melting Range or Temperature á741ñ).
(see Melting Range or Temperature á741ñ).
B:
Infrared Absorption á197Kñ:using a portion of the dried precipitate obtained in Identificationtest A.
Specific rotation á781Sñ:
between –137 and –149
and –149 .
.
Test solution:
25mg per mL,in water,calculated on the anhydrous,solvent-free basis.
Water,Method Iá921ñ:
not more than 7.0%.
Residue on ignition á281ñ:
not more than 0.05%,the use of sulfuric acid being omitted.
Limit of alcohol (C2H5OH)—
Internal standard stock solution—
Transfer 6.0mLof isopropyl alcohol to a 500-mLvolumetric flask,dilute with water to volume,and mix.[NOTE—The isopropyl alcohol must be free of alcohol impurities.]
Internal standard solution—
Transfer 5.0mLof Internal standard stock solutionto a 100-mLvolumetric flask,dilute with water to volume,and mix.
Standard solution—
Transfer 2mLof alcohol to a 100-mLvolumetric flask,dilute with water to volume,and mix.Pipet 3.0mLof this solution and 5.0mLof the Internal standard stock solutioninto a 100-mLvolumetric flask,dilute with water to volume,and mix.
Test solution—
Transfer about 240mg of Oxycodone Hydrochloride,accurately weighed,to a 15-mLcentrifuge tube,add 5.0mLof Internal standard solution,and mix to dissolve.
Chromatographic system (seeChromatography á621ñ)—
The gas chromatograph is equipped with a flame-ionization detector and a 4-mm ×1.8-m glass column that is packed with 80-to 100-mesh support S3,helium being used as the carrier gas.Prior to use,condition the column overnight at 235 with a slow flow of carrier gas.The column is maintained at 150
with a slow flow of carrier gas.The column is maintained at 150 ,and the injection port and detector temperatures are maintained at 170
,and the injection port and detector temperatures are maintained at 170 .Chromatograph the Standard solution,and record the peak responses as directed for Procedure:the resolution,R,between isopropyl alcohol and alcohol is not less than 2;the tailing factor for alcohol is not more than 1.5;and the relative standard deviation for replicate injections is not more than 2.0%.
.Chromatograph the Standard solution,and record the peak responses as directed for Procedure:the resolution,R,between isopropyl alcohol and alcohol is not less than 2;the tailing factor for alcohol is not more than 1.5;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 5µL)of the Standard solutionand the Test solutioninto the chromatograph,record the chromatograms,and measure the peak responses for the major peaks.Calculate the percentage of C2H5OHin the portion of Oxycodone Hydrochloride taken by the formula:
(240/W)(RU/RS),
in which Wis the weight,in mg,of Oxycodone Hydrochloride in the Test solution;and RUand RSare the ratios of the alcohol peak to the isopropyl alcohol peak obtained from the Test solutionand the Standard solution,respectively.[NOTE—The density of alcohol,800mg per mL,has been accounted for in the calculations.]Not more than 1.0%(w/w)of alcohol (C2H5OH)is found.
Chloride content—
Dissolve about 300mg,accurately weighed,in 50mLof methanol,add 5mLof glacial acetic acid,and titrate with 0.1Nsilver nitrate VS,determining the endpoint potentiometrically.Each mLof 0.1Nsilver nitrate is equivalent to 3.545mg of Cl:the content of Cl is between 9.8%and 10.4%,calculated on the anhydrous,solvent-free basis.
Chromatographic purity—
Using the chromatogram of the Assay preparationobtained in the Assay,calculate the percentage of each impurity in the Oxycodone Hydrochloride taken by the formula:
100(ri/rs),
in which riis the peak response for each impurity;and rsis the sum of the responses of all of the peaks:not more than 1.0%of any individual impurity is found;and the sum of all impurities is not more than 2.0%.
Assay—
Mobile phase—
Prepare a mixture of 0.005Msodium 1-hexanesulfonate,methanol,phosphoric acid,and triethylamine (900:100:5:2).Adjust with 50%sodium hydroxide solution to a pHof 2.5±0.1,filter,and degas.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Resolution solution—
Prepare a solution in Mobile phasecontaining about 13µg of codeine phosphate and 9µg of oxycodone per mL.
Standard preparation—
Dissolve an accurately weighed quantity of USP Oxycodone RSquantitatively in Mobile phaseto obtain a solution having a known concentration of about 0.9mg per mL.
Assay preparation—
Transfer about 100mg of Oxycodone Hydrochloride,accurately weighed,to a 100-mLvolumetric flask,add about 50mLof Mobile phase,and swirl to dissolve.Dilute with Mobile phaseto volume,and mix.Filter a portion of this solution through a filter having a porosity of 0.5µm or finer,and use the filtrate as the Assay preparation.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 206-nm detector and a 3.9-mm ×15-cm column that contains 4-µm packing L7and is maintained at a constant temperature of about 50 .The flow rate is about 1.5mLper minute.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 0.8for codeine and 1.0for oxycodone;and the resolution,R,between the codeine peak and the oxycodone peak is not less than 3.0.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the tailing factor is between 0.75and 1.25;and the relative standard deviation for replicate injections is not more than 2.0%.
.The flow rate is about 1.5mLper minute.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 0.8for codeine and 1.0for oxycodone;and the resolution,R,between the codeine peak and the oxycodone peak is not less than 3.0.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the tailing factor is between 0.75and 1.25;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms for a period of time that is twice the retention time of the main oxycodone peak,and measure the responses for all the peaks.Calculate the quantity,in mg,of C18H21NO4·HCl in the portion of Oxycodone Hydrochloride taken by the formula:
(351.82/315.37)(100C)(rU/rS),
in which 351.82and 315.37are the molecular weights of oxycodone hydrochloride and oxycodone base,respectively;Cis the concentration,in mg per mL,of USP Oxycodone RSin the Standard preparation;and rUand rSare the oxycodone peak area responses obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Daniel K.Bempong,Ph.D.,Scientist
Expert Committee:(PA2)Pharmaceutical Analysis 2
USP28–NF23Page 1436
Pharmacopeial Forum:Volume No.28(1)Page 84
Phone Number:1-301-816-8143