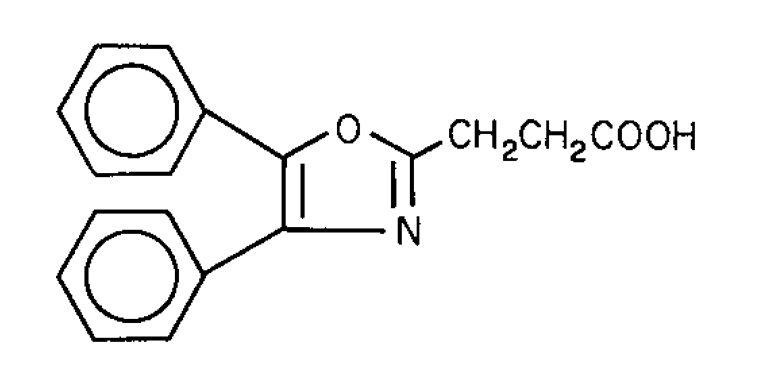
Oxaprozin
2-Oxazolepropanoic acid,4,5-diphenyl-.
4,5-Diphenyl-2-oxazolepropionic acid [21256-18-8].
»Oxaprozin contains not less than 98.5percent and not more than 101.5percent of C18H15NO3,calculated on the dried basis.
NOTE—Because of light sensitivity,protect all oxaprozin samples and Standard solutions from light.
Identification—
A:
Infrared Absorption á197Kñ:previously dried at 105 for 2hours.
for 2hours.
B:
Ultraviolet Absorption á197Uñ:previously dried at 105 for 2hours.The absorbance of the sample at 285nm is between 0.455and 0.495.
for 2hours.The absorbance of the sample at 285nm is between 0.455and 0.495.
Solution:
10µg per mL.
Medium:
methanol.
Loss on drying á731ñ—
Dry it at 105 for 2hours:it loses not more than 0.3%of its weight.
for 2hours:it loses not more than 0.3%of its weight.
Residue on ignition á281ñ:
not more than 0.3%.
Arsenic,Method IIá211ñ:
1µg per g.
Heavy metals,Method IIá231ñ:
0.001%.
Chromatographic purity—
Solution A:
0.1%phosphoric acid adjusted with phosphoric acid to a pHof 2.00±0.1.
Solution B—
Use acetonitrile.
Mobile phase—
Use variable mixtures of Solution Aand Solution Bas directed for Chromatographic system.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Diluent A:
a mixture of acetonitrile,methylene chloride,and water (48:1:1).
Diluent B:
a mixture of acetonitrile and water (1:1).
Standard stock solution—
Dissolve an accurately weighed quantity of USP Oxaprozin RSin acetonitrile to obtain a solution having a concentration of about 200µg per mL.
Standard solution—
Transfer 5.0mLof Standard stock solutionto a 200-mLvolumetric flask,and dilute with Diluent Ato volume.
Benzil solution:
200µg of benzil per mLin acetonitrile.
Resolution solution—
Transfer 5.0mLof Benzil solutionand 5.0mLof Standard stock solutionto a 100-mLvolumetric flask,and dilute with Diluent Ato volume to obtain a solution having known concentrations of about 10µg of each per mL.
Test solution A—
[NOTE—Test solution Ais used to monitor all known and unknown impurities,except imidazolic acid and oximide.]Transfer about 100mg of Oxaprozin,accurately weighed,to a 100-mLvolumetric flask;add 2mLof methylene chloride,2mLof water,and 75mLof acetonitrile;and sonicate after each solvent is added.Dilute with acetonitrile to volume.
Test solution B—
[NOTE—Test solution Bis used to monitor only imidazolic acid and oximide.]Transfer about 100mg of Oxaprozin,accurately weighed,to a 100-mLvolumetric flask;add 75mLof Diluent Bto dissolve the Oxaprozin;and dilute withDiluent Bto volume.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 238-nm detector and a 4.6-mm ×15-cm column that contains 5-µm packing L7.The flow rate is 1.0mLper minute.The chromatograph is programmed as follows.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
| 0 | 70 | 30 | equilibration |
| 0–20 | 70 | 30 | isocratic |
| 21–60 | 70®0 | 30®100 | linear gradient |
| 60–61 | 0®70 | 100®30 | linear gradient |
| 61–70 | 70 | 30 | isocratic |
Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 1.1for benzil and 1.0for oxaprozin;and the resolution,R,between oxaprozin and benzil is not less than 3.0.Chromatograph the Standard solution,and record the peak responses as directed for Procedure:the tailing factor is not more than 2.0,and the relative standard deviation for replicate injections is not more than 5.0%.
Procedure—
Inject 20µLof Test solution Aand Test solution Binto the chromatograph,record the chromatogram,and measure the areas for all the peaks.Calculate the percentage of each impurity in the portion of Oxaprozin taken by the formula:
100(Fri/rs),
in which Fis the relative response factor and is equal to 1.15for the imidazolic acid peak with a relative retention time of 0.14,1.21for any peak with a relative retention time of 0.42,0.91for the oximide peak with a relative retention time of 0.73,0.85for any peak with a relative retention time of 0.84,1.29for any peak with a relative retention time of 1.08,1.46for any peak with a relative retention time of 1.50,and 2.09for any peak with a relative retention time of 1.57;riis the peak response for each impurity;and rsis the sum of the responses of all the peaks:not more than 0.1%of any individual impurity is found,and not more than 0.5%of total impurities is found.[NOTE—The values of Ffor all known impurities except imidazolic acid and oximide were found using Test solution A,and the values of Ffor imidazolic acid and oximide were found using Test solution B.]
Organic volatile impurities,Method Vá467ñ:
meets the requirements.
Solvent—
Use dimethyl sulfoxide.
Assay—
Dissolve about 400mg of Oxaprozin,previously dried at 105 for 2hours and accurately weighed,in about 100mLof alcohol in a narrow-mouth container,and titrate with 0.1Nsodium hydroxide VS,determining the endpoint potentiometrically (see Titrimetry á541ñ).Each mLof 0.1Nsodium hydroxide is equivalent to 29.332mg of oxaprozin.
for 2hours and accurately weighed,in about 100mLof alcohol in a narrow-mouth container,and titrate with 0.1Nsodium hydroxide VS,determining the endpoint potentiometrically (see Titrimetry á541ñ).Each mLof 0.1Nsodium hydroxide is equivalent to 29.332mg of oxaprozin.
Auxiliary Information—
Staff Liaison:Clydewyn M.Anthony,Ph.D.,Scientist
Expert Committee:(PA2)Pharmaceutical Analysis 2
USP28–NF23Page 1427
Pharmacopeial Forum:Volume No.29(4)Page 1059
Phone Number:1-301-816-8139