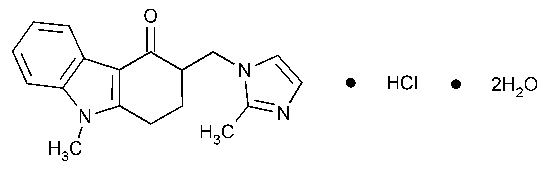
Ondansetron Hydrochloride
4H-Carbazol-4-one,1,2,3,9-tetrahydro-9-methyl-3-(2-methyl-1H-imidazol-1-yl)methyl-,monohydrochloride,(±)-,dihydrate.
(±)-2,3-Dihydro-9-methyl-3-(2-methylimidazol-1-yl)methylcarbazol-4(1H)-one monohydrochloride dihydrate [103639-04-9].
»Ondansetron Hydrochloride contains not less than 98.0percent and not more than 102.0percent of C18H19N3O·HCl,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight,light-resistant containers.Store at 25 ,excursions permitted between 15
,excursions permitted between 15 and 30
and 30 .
.
USP Reference standards á11ñ—
USP Ondansetron Hydrochloride RS.USP Ondansetron Related Compound A RS.USP Ondansetron Related Compound B RS.USP Ondansetron Related Compound C RS.USP Ondansetron Related Compound D RS.
Identification—
A:Infrared Absorption á197Mñ.
B:
Dissolve 20mg in 2mLof water,add 1mLof 2Mnitric acid,and filter:the filtrate responds to the test for Chloride á191ñ.
Water,Method Ia á921ñ:
between 9.0%and 10.5%.
Residue on ignition á281ñ:
not more than 0.1%.
Limit of ondansetron related compound D—
Mobile phase—
Prepare a filtered and degassed mixture of 0.02Mmonobasic potassium phosphate (previously adjusted with 1Msodium hydroxide to a pHof 5.4)and acetonitrile (80:20).Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard solution—
Dissolve an accurately weighed quantity of USP Ondansetron Related Compound D RSin Mobile phase;and dilute quantitatively,and stepwise if necessary,with Mobile phaseto obtain a solution having a known concentration of about 0.4µg per mL.
System suitability solution—
Dissolve suitable quantities of USP Ondansetron Related Compound D RSand USP Ondansetron Related Compound C RSin Mobile phase;and dilute quantitatively,and stepwise if necessary,with Mobile phaseto obtain a solution having a concentration of about 0.6µg per mLand 1µg per mL,respectively.
Test solution—
Transfer about 50mg of Ondansetron Hydrochloride,accurately weighed,to a 100-mLvolumetric flask,dissolve in and dilute with Mobile phaseto volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 328-nm detector and a 4.6-mm ×20-cm column that contains packing L10.The flow rate is about 1.5mLper minute.Chromatograph the System suitability solution,and record the peak responses as directed forProcedure:the relative retention times are about 0.8for ondansetron related compound Cand 1.0for ondansetron related compound D;and the resolution,R,between ondansetron related compound Cand ondansetron related compound Dis not less than 1.5.Chromatograph the Standard solution,and record the peak responses as directed forProcedure:the column efficiency determined from the analyte peak is not less than 400theoretical plates;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 20µL)of the Standard solutionand the Test solutioninto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the percentage of ondansetron related compound Din the portion of Ondansetron Hydrochloride taken by the formula:
10(C/W)(rU/rS),
in which Cis the concentration,in µg per mL,of USP Ondansetron Related Compound D RSin the Standard solution;Wis the weight,in mg,of Ondansetron Hydrochloride taken to prepare the Test solution;and rUand rSare the peak responses obtained from the Test solutionand the Standard solution,respectively:not more than 0.1%is found.
Chromatographic purity—
METHOD I—
Resolution solution—
Dissolve an accurately weighed quantity of USP Ondansetron Related Compound A RSin methanol;and dilute quantitatively,and stepwise if necessary,with methanol to obtain a solution having a known concentration of 100µg per mL.
Identification solution—
Dissolve an accurately weighed quantity of USP Ondansetron Related Compound B RSin methanol;and dilute quantitatively,and stepwise if necessary,with methanol to obtain a solution having a known concentration of 100µg per mL.
Standard solutions—
Dissolve an accurately weighed quantity of USP Ondansetron Hydrochloride RSin methanol,and mix to obtain a solution having a known concentration of about 0.25mg per mL.Quantitatively dilute this solution with methanol to obtain Standard solutions,designated below by letter,having the following compositions:
| Standard solution |
Dilution | Concentration (µg RS per mL) |
Percentage (%,for comparison with test specimen) |
| A | (1in 5) | 50 | 0.4 |
| B | (1in 10) | 25 | 0.2 |
| C | (1in 20) | 12.5 | 0.1 |
Test solution—
Dissolve an accurately weighed quantity of Ondansetron Hydrochloride in methanol to obtain a solution containing 12.5mg per mL.
Procedure—
Separately apply 20µLof the Test solution,20µLof each Standard solution,and 10µLof the Identification solutionto a thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel mixture.To the same plate apply 20µLof the Test solution,and on top of this application apply 10µLof the Resolution solutionand 10µLof the Identification solutionto make a system suitability spot.Develop the chromatogram in a solvent system consisting of a mixture of chloroform,ethyl acetate,methanol,and ammonium hydroxide (90:50:40:1)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the chamber,mark the solvent front,and allow the solvent to evaporate.Examine the plate under short-wavelength UVlight,and compare the intensities of any secondary spots observed in the chromatogram of the Test solutionwith those of the principal spots in the chromatograms of the Standard solutions:complete resolution of the three components of the system suitability spot,any secondary spot from the chromatogram of the Test solutionhaving an RFvalue corresponding to that of the principal spot of the Identification solutionis not larger or more intense than the principal spot obtained from the Standard solution A(0.4%);and no other secondary spot from the chromatogram of the Test solution having an RFvalue corresponding to that of the principal spot of the Resolution solutionis larger or more intense than the principal spot obtained from Standard solution B(0.2%).
METHOD II—
Mobile phaseand Chromatographic system—
Proceed as directed in the Assay.
Standard solution—
Proceed as directed for Standard preparationin the Assay.
Test solution—
Use the Assay preparation.
Procedure—
Separately inject equal volumes (about 10µL)of the Standard solutionand the Test solutioninto the chromatograph,record the chromatogram,and measure the peak responses.Calculate the percentage of each impurity in the portion of Ondansetron Hydrochloride taken by the formula:
50,000(C/W)(1/F)(ri/rS),
in which Cis the concentration,in mg per mL,of USP Ondansetron Hydrochloride RSin the Standard solution;Wis the weight,in mg,of Ondansetron Hydrochloride taken to prepare the Test solution;Fis the relative response factor of the impurities as described in the accompanying table;riis the peak area for each impurity in the Test solution;and rSis the peak area of ondansetron obtained from the Standard solution:meets the requirements given in the accompanying table.
| Compound name | Relative retention time | Relative response factor | Limit (%) |
| Ondansetron related compound C | about 0.32 | 1.2 | 0.2 |
| Ondansetron related compound D* | about 0.34 | 1.3 | 0.1 |
| Imidazole | about 0.49 | 0.3 | 0.2 |
| 2-methylimidazole | about 0.54 | 0.4 | 0.2 |
| Ondansetron | 1.0 | — | — |
| Ondansetron related compound A | about 1.10 | 0.8 | 0.2 |
| Unknown | — | 1.0 | 0.1 |
| Total | — | — | 0.5 |
|
*
Quantified in the test for Limit of ondansetron related compound D.
|
|||
Assay—
Mobile phase—
Prepare a filtered and degassed mixture of 0.02Mmonobasic sodium phosphate (previously adjusted with 1Msodium hydroxide to a pHof 5.4)and acetonitrile (50:50).Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Ondansetron Hydrochloride RSin Mobile phase;and dilute quantitatively,and stepwise if necessary,with Mobile phaseto obtain a solution having a known concentration of about 90µg per mL.
System suitability solution—
Dissolve suitable quantities of USP Ondansetron Hydrochloride RSand USP Ondansetron Related Compound A RSin Mobile phase;and dilute quantitatively,and stepwise if necessary,with Mobile phaseto obtain a solution containing about 90µg per mLand 20µg per mL,respectively.
Assay preparation—
Transfer about 45mg of Ondansetron Hydrochloride,accurately weighed,to a 50-mLvolumetric flask;dissolve in and dilute with Mobile phaseto volume;and mix.Pipet 5.0mLof this solution into a 50-mLvolumetric flask,dilute with Mobile phaseto volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 216-nm detector and a 4.6-mm ×20-cm column that contains packing L10.The flow rate is about 1.5mLper minute.Chromatograph the System suitability solution,and record the peak responses as directed forProcedure:the relative retention times are about 1.0for ondansetron and 1.1for ondansetron related compound A;and the resolution,R,between ondansetron related compound Aand ondansetron is not less than 1.5.Chromatograph the Standard preparation,and record the peak responses as directed forProcedure:the tailing factor is not more than 2.0;and the relative standard deviation for replicate injections is not more than 1.5%.
Procedure—
Separately inject equal volumes (about 10µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in mg,of C18H19N3O·HCl in the portion of Ondansetron Hydrochloride taken by the formula:
500C(rU/rS),
in which Cis the concentration,in mg per mL,of USP Ondansetron Hydrochloride RSin the Standard preparation;and rUand rSare the peak responses obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Salvador C.Salado,M.S.,Scientist and Latin American Liaison
Expert Committee:(PA3)Pharmaceutical Analysis 3
USP28–NF23Page 1418
Pharmacopeial Forum:Volume No.29(6)Page 1941
Phone Number:1-301-816-8165