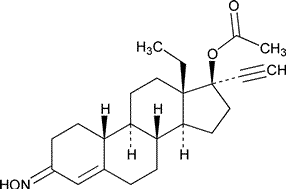
Norgestimate
»Norgestimate is a mixture of (E)-and(Z)-isomers having a ratio of (E)-to (Z)-isomer between 1.27and 1.78and it contains not less than 98.0percent and not more than 102.0percent of C23H31NO3,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
A:Infrared Absorption á197Kñ—
Test specimen—
Use a dispersion in potassium bromide prepared by mixing the specimen with potassium bromide in a 1to 100ratio.
B:
The retention time of the major peak in the chromatogram of theAssay preparation corresponds to that in the chromatogram of theStandard preparation,as obtained in the Assay.
Specific rotation á781Sñ:
between +40 and +46
and +46 .
.
Test solution:
10mg per mL,in chloroform.
Loss on drying á731ñ—
Dry it at 105 for 3hours:it loses not more than 0.5%of its weight.
for 3hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.3%.
Heavy metals,Method IIá231ñ:
0.002%.
Limit of residual solvents—
Internal standard solution—
Prepare a solution of isobutyl alcohol in dimethylformamide containing 2µLof isobutyl alcohol per 100mLof solution.
Standard solution—
Prepare a solution inInternal standard solution containing 5µLeach of acetone,alcohol,chloroform,diisopropyl ether,and methanol per 100mLof solution.
System suitability solution—
Dilute a portion of theStandard solution withInternal standard solution to obtain a solution containing 0.05µLeach of acetone,alcohol,chloroform,diisopropyl ether,and methanol per 100mLof solution.
Test solution—
Transfer about 40mg of Norgestimate and 2mLofInternal standard solution to a 5-mLvolumetric flask or a suitable vial,and shake well to dissolve.
Chromatographic system(see Chromatography á621ñ)—
The gas chromatograph is equipped with a flame-ionization detector,a 0.53-mm ×30-m fused-silica capillary column bonded with a 1-µm layer of phase G16,and a split injection system.The detector temperature is about 250 ,and the injection port temperature is about 180
,and the injection port temperature is about 180 .The column temperature is programmed as follows.It is maintained at about 65
.The column temperature is programmed as follows.It is maintained at about 65 for 2.5minutes,increased at a rate of 35
for 2.5minutes,increased at a rate of 35 per minute to 100
per minute to 100 ,maintained for 2minutes,then at a rate of 30
,maintained for 2minutes,then at a rate of 30 per minute increased to 160
per minute increased to 160 ,and maintained for 2.5minutes.The carrier gas is helium,flowing at a rate of about 6mLper minute,and the split flow rate is about 16mLper minute.Chromatograph theInternal standard solution,theStandard solution,and theSystem suitability solution,and record the peak responses as directed forProcedure:there are no interfering peaks due to dimethylformamide;the retention time of isobutyl alcohol in the chromatogram of theInternal standard solution is between 4and 5minutes;the signal-to-noise ratio for alcohol obtained from theSystem suitability solution is not less than 2.0;and the relative standard deviation for replicate injections of theStandard solution,determined from the peak response ratios of each solvent to the internal standard,is not more than 3.0%.
,and maintained for 2.5minutes.The carrier gas is helium,flowing at a rate of about 6mLper minute,and the split flow rate is about 16mLper minute.Chromatograph theInternal standard solution,theStandard solution,and theSystem suitability solution,and record the peak responses as directed forProcedure:there are no interfering peaks due to dimethylformamide;the retention time of isobutyl alcohol in the chromatogram of theInternal standard solution is between 4and 5minutes;the signal-to-noise ratio for alcohol obtained from theSystem suitability solution is not less than 2.0;and the relative standard deviation for replicate injections of theStandard solution,determined from the peak response ratios of each solvent to the internal standard,is not more than 3.0%.
Procedure—
Separately inject equal volumes (about 1µL)of theStandard solution and theTest solution into the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the percentage of each solvent in the portion of Norgestimate taken by the formula:
200(CD/W)(RU/RS),
in whichCis the concentration,in mLper mL,of each solvent in theStandard solution;Dis the density,in mg per mL,of each solvent;Wis the weight,in mg,of Norgestimate taken to prepare theTest solution;andRUandRSare the peak response ratios of the appropriate analyte to the internal standard obtained from theTest solution and theStandard solution,respectively.Not more than 0.5%each of acetone and alcohol is found;not more than 0.05%of diisopropyl ether is found;not more than 0.006%of chloroform is found;and not more than 0.3%of methanol is found.
Chromatographic purity—
TEST1—
Diluent,Mobile phase,System suitability solution,and Chromatographic system—
Proceed as directed in theAssay.
Standard solution—
Use theStandard preparation,prepared as directed in theAssay.
Test solution—
Use theAssay preparation,prepared as directed in theAssay.
Procedure—
Separately inject equal volumes (about 25µL)of theStandard solution and theTest solution into the chromatograph,record the chromatograms,and measure the peak areas.Calculate the percentage of each impurity in the portion of Norgestimate taken by the formula:
5000(CP/W)(ri/FrS),
in whichCis the concentration,in mg per mL,of USP Norgestimate RSin theStandard solution;Pis the fraction of (E)-norgestimate in USP Norgestimate RS;Wis the weight,in mg,of Norgestimate taken to prepare theTest solution;riis the peak area for each impurity obtained from theTest solution;Fis the relative response factor and it is equal to 0.83for any peak having a relative retention time of 0.50,1.13for any peak having a relative retention time of 0.56,0.85for any peak having a relative retention time of 0.72,and 1.0for any other peak;andrSis the peak area of (E)-norgestimate obtained from theStandard solution.Not more than 0.3%of total impurities having relative retention times of 0.50and 0.56is found;not more than 0.3%of the impurity having a relative retention time of 0.72is found;and not more than 0.1%of any other impurity is found.
TEST2—
Mobile phase—
Prepare a filtered and degassed mixture of cyclohexane and absolute alcohol (50:1).Make adjustments if necessary (seeSystem Suitability underChromatography á621ñ).
Standard solution—
Dissolve an accurately weighed quantity of USP Norgestimate RSinMobile phase,and dilute quantitatively,and stepwise if necessary,withMobile phase to obtain a solution having a known concentration of about 1.0mg per mL.
System suitability solution—
Dilute a portion of theStandard solution,quantitatively and stepwise if necessary,withMobile phase to obtain a solution having a known concentration of about 0.5µg per mL.
Test solution—
Transfer about 10mg of Norgestimate,accurately weighed,to a 10-mLvolumetric flask,dissolve in and dilute withMobile phase to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 210-nm detector and a 4.6-mm ×25-cm column that contains a 5-µm packing L20.The flow rate is about 1mLper minute.Chromatograph theSystem suitability solution,and record the peak responses as directed forProcedure:the signal-to-noise ratio for (E)-norgestimate is not less than 3.0.Chromatograph theStandard solution,and record the peak areas as directed forProcedure:the retention time is about 18.6minutes for(E)-norgestimate;the relative retention times are about 1.0for(E)-norgestimate and 1.1for (Z)-norgestimate;the tailing factor is not more than 1.5;the resolution,R,between (Z)-norgestimate and (E)-norgestimate is not less than 1.5;and the relative standard deviation for replicate injections,determined from the peak area of(Z)-norgestimate to(E)-norgestimate,is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 25µL)of theStandard solution and theTest solution into the chromatograph,record the chromatograms,and measure the peak areas.Calculate the percentage of each impurity in the portion of Norgestimate taken by the formula:
1000(CP/W)(ri/FrS),
in whichCis the concentration,in mg per mL,of USP Norgestimate RSin theStandard solution;Pis the fraction of(E)-norgestimate in USP Norgestimate RS;Wis the weight,in mg,of Norgestimate taken to prepare theTest solution;riis the peak area for each impurity obtained from theTest solution;Fis the relative response factor and it is equal to 1.4for any peak having a relative retention time of 0.74,1.5for any peak having a relative retention time of 0.78,and 1.2for any peak having a relative retention time of 0.91;andrSis the peak area of(E)-norgestimate obtained from theStandard solution.Not more than 0.2%of the impurity having a relative retention time of 0.74is found;and not more than 0.1%each of the impurities having relative retention times of 0.78and 0.91.Not more than 1.0%of total impurities is found,the results forTest 1andTest 2being added.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.Chloroform is tested in theLimit of residual solvents test.
Solvent—
Use dimethyl sulfoxide.
Assay—
Diluent—
Prepare a mixture of water and methanol (1:4).
Mobile phase—
Prepare a filtered and degassed mixture of water,tetrahydrofuran,and acetonitrile (30:11:9).Make adjustments if necessary (seeSystem Suitability underChromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Norgestimate RSinDiluent,and dilute quantitatively,and stepwise if necessary,withDiluent to obtain a solution having a known concentration of about 0.5mg per mL.
System suitability solution—
Dilute a portion ofStandard preparation,quantitatively and stepwise if necessary,with Diluent to obtain a solution having a known concentration of about 0.05µg per mL.
Assay preparation—
Transfer about 25mg of Norgestimate,accurately weighed,to a 50-mLvolumetric flask,dissolve in and dilute withDiluent to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 244-nm detector and a 4.6-mm ×10-cm column that contains a 3-µm packing L1.The flow rate is about 1.2mLper minute.The column temperature is maintained at about 40 .Chromatograph theSystem suitability solution,and record the peak areas as directed for Procedure:the signal-to-noise ratio for(Z)-norgestimate is not less than 3.0.Chromatograph theStandard preparation,and record the peak areas as directed forProcedure:the relative retention times are about 0.86for(Z)-norgestimate and 1.0for (E)-norgestimate;the resolution,R,between (Z)-norgestimate and(E)-norgestimate is not less than 1.5;the tailing factor for (E)-norgestimate and for(Z)-norgestimate is not more than 1.5;and the relative standard deviation for replicate injections,determined from the peak area ratio of(E)-norgestimate to (Z)-norgestimate,is not more than 2.0%.
.Chromatograph theSystem suitability solution,and record the peak areas as directed for Procedure:the signal-to-noise ratio for(Z)-norgestimate is not less than 3.0.Chromatograph theStandard preparation,and record the peak areas as directed forProcedure:the relative retention times are about 0.86for(Z)-norgestimate and 1.0for (E)-norgestimate;the resolution,R,between (Z)-norgestimate and(E)-norgestimate is not less than 1.5;the tailing factor for (E)-norgestimate and for(Z)-norgestimate is not more than 1.5;and the relative standard deviation for replicate injections,determined from the peak area ratio of(E)-norgestimate to (Z)-norgestimate,is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 25µL)of theStandard preparation and theAssay preparation into the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the quantity,in mg,of C23H31NO3in the portion of Norgestimate taken by the formula:
50C(rU/rS),
in whichCis the concentration,in mg per mL,of USP Norgestimate RSin theStandard preparation;andrUandrSare the sums of peak areas of (Z)-norgestimate and (E)-norgestimate obtained from theAssay preparation and theStandard preparation,respectively.Calculate the percentages of the(Z)-and (E)-isomers,UZand UE,respectively,in the portion of Norgestimate taken by the formula:
5000(CP/W)(rU/rS),
in whichCis the concentration,in mg per mL,of USP Norgestimate RSin theStandard preparation;Pis the fraction of(E)-or(Z)-norgestimate in USP Norgestimate RS;Wis the weight,in mg,of Norgestimate taken to prepare theAssay preparation;andrUandrSare the peak responses of the appropriate norgestimate isomer obtained from theAssay preparation and theStandard preparation,respectively.Calculate the ratio of (E)-norgestimate to (Z)-norgestimate,that is,the ratio of UEto UZ.
Auxiliary Information—
Staff Liaison:Clydewyn M.Anthony,Ph.D.,Scientist
Expert Committee:(PA1)Pharmaceutical Analysis 1
USP28–NF23Page 1401
Pharmacopeial Forum:Volume No.29(6)Page 1938
Phone Number:1-301-816-8139