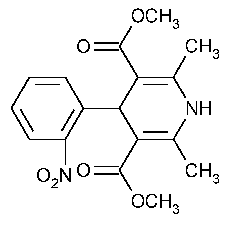
Nifedipine
»Nifedipine contains not less than 98.0percent and not more than 102.0percent of C17H18N2O6,calculated on the dried basis.
Packaging and storage—
Preserve in tight,light-resistant containers.
USP Reference standards á11ñ—
USP Nifedipine RS.USP Nifedipine Nitrophenylpyridine Analog RS.USP Nifedipine Nitrosophenylpyridine Analog RS.
NOTE—Nifedipine,when exposed to daylight and certain wavelengths of artificial light,readily converts to a nitrosophenylpyridine derivative.Exposure to UVlight leads to the formation of a nitrophenylpyridine derivative.Perform assays and tests in the dark or under golden fluorescent or other low-actinic light.Use low-actinic glassware.
Identification—
A:
Infrared Absorption á197Kñ—Do not dry specimens.
B:Ultraviolet Absorption á197Uñ—
Spectral range:
450to 200nm.
Solution—
To a 10-mLvolumetric flask containing 14mg of Nifedipine add 1.0mLof chloroform,dilute with methanol to volume,and mix.Pipet a 1.0-mLaliquot of the solution into a 100-mLvolumetric flask,dilute with methanol to volume,and mix.
Medium:
methanol.
C:
The retention time of the major peak in the chromatogram of the Assay preparationcorresponds to that in the chromatogram of the Standard preparation,as obtained in the Assay.
Loss on drying á731ñ—
Dry it at 105 to constant weight:it loses not more than 0.5%of its weight.
to constant weight:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%,an ignition temperature of 600 being used.
being used.
Heavy metals,Method IIá231ñ:
0.001%.
Perchloric acid titration—
Transfer about 4g of Nifedipine,accurately weighed,to a 250-mLconical flask,and dissolve in 160mLof glacial acetic acid with the aid of an ultrasonic bath.Add 3drops of p-naphtholbenzein TS,and titrate to a green endpoint with 0.1Nperchloric acid VS:not more than 0.12mLof 0.1Nperchloric acid is consumed for each g of Nifedipine.
Chloride and Sulfate—
To 5.0g of Nifedipine in a 140-mLbeaker add 4.0mLof 6Nacetic acid and 46mLof water.Bring carefully to a boil on a hot plate,cool,and filter through paper free of chloride and sulfate.Use this Nifedipine filtrate for the following tests.
Chloride—
Pipet 2.5mLof the Nifedipine filtrate into a 50-mLcolor-comparison tube,and add 12.5mLof water.Into a matched color-comparison tube pipet 10mLof a Standard solution containing 8.2µg of sodium chloride per mLcorresponding to 5µg of chloride per mL,add 5.0mLof water,and mix.To each tube add 0.15mLof 0.3Mnitric acid and 0.3mLof silver nitrate TS,and mix:the opalescence exhibited by the Nifedipine filtrate does not exceed that of the Standard solution (0.02%).
Sulfate—
Pipet into each of two 50-mLmatched color-comparison tubes 1.5mLof sulfate solution consisting of sufficient potassium sulfate dissolved in water to obtain a sulfate concentration of 10µg per mL.To each tube add,successively and with continuous shaking,0.75mLof alcohol,0.5mLof a 6.1%aqueous solution of barium chloride,and 0.25mLof 6Nacetic acid.Shake for an additional 30seconds.Pipet into one tube,designated the Standard tube,15mLof the sulfate solution.Pipet into the other tube,designated the Specimen tube,3mLof the Nifedipine filtrate and 12mLof water:the turbidity exhibited by the Specimen tube does not exceed that of the Standard tube (0.05%).
Related compounds—
[NOTE—Protect the Standard nifedipine solutionand the Test preparationfrom actinic light.Conduct this test promptly after preparation of the Standard nifedipine solutionand the Test solution.]
Mobile phase—
Prepare as directed in the Assay.
Standard nifedipine solution—
Dissolve an accurately weighed quantity of USP Nifedipine RSin methanol (about 1mg per mL),and dilute quantitatively with Mobile phaseto obtain a solution having a known concentration of about 0.3mg per mL.
Reference solution 1—
Dissolve an accurately weighed quantity of USP Nifedipine Nitrophenylpyridine Analog RSin methanol (about 1mg per mL),and dilute quantitatively with Mobile phaseto obtain a solution having a known concentration of about 0.6µg per mL.
Reference solution 2—
Dissolve an accurately weighed quantity of USP Nifedipine Nitrosophenylpyridine Analog RSin methanol (about 1mg per mL),and dilute quantitatively with Mobile phaseto obtain a solution having a known concentration of about 0.6µg per mL.
Standard solution—
Transfer 5.0mLof each of the two Reference solutionsto a container,add 5.0mLof Mobile phase,and mix.
Test solution—
Prepare as directed for the Assay preparationin the Assay.
System suitability solution—
Mix equal volumes of the Standard nifedipine solutionand of each of the two Reference solutions.
Chromatographic system—
Prepare as directed in the Assay.Chromatograph the System suitability preparation,and record the peak responses as directed for Procedure:the resolution,R,between the nitrophenylpyridine analog and nitrosophenylpyridine analog peaks is not less than 1.5;the resolution,R,between the nitrosophenylpyridine analog and nifedipine peaks is not less than 1.0;and the relative standard deviation of the response for each analog in replicate injections is not more than 10%.The relative retention times are about 0.8for the nitrophenylpyridine analog,about 0.9for the nitrosophenylpyridine analog,and 1.0for nifedipine.
Procedure—
Separately inject equal volumes (about 25µL)of the Standard solutionand the Test solutioninto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in mg,of each related compound in the portion of Nifedipine taken by the formula:
250C(rU/rS),
in which Cis the concentration,in mg per mL,of the appropriate USP Nifedipine Analog RS,in the Standard solution;and rUand rSare the peak responses for the corresponding related compound obtained from the Test solutionand the Standard solution,respectively.Not more than 0.2%of each of dimethyl 4-(2-nitrophenyl)-2,6-dimethylpyridine-3,5-dicarboxylate and dimethyl-4-(2-nitrosophenyl)-2,6-dimethylpyridine-3,5-dicarboxylate,corresponding to Nifedipine Nitrophenylpyridine Analog and Nifedipine Nitrosophenylpyridine Analog,respectively,is found.
Organic volatile impurities,Method Vá467ñ:
meets the requirements.
Solvent:
dimethyl sulfoxide.
Assay—
[NOTE—Protect the Standard preparationand the Assay preparationfrom actinic light.Conduct the Assaypromptly after preparation of the Standard preparationand the Assay preparation.]
Mobile phase—
Prepare a suitable mixture of water,acetonitrile,and methanol (50:25:25),and degas.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Nifedipine RSin methanol (about 1mg per mL),and quantitatively dilute with Mobile phaseto obtain a solution having a known concentration of about 0.1mg per mL.
Assay preparation—
Transfer about 25mg of Nifedipine,accurately weighed,to a 250-mLvolumetric flask.Dissolve in 25mLof methanol,dilute with Mobile phaseto volume,and mix to obtain a solution having a concentration of about 0.1mg per mL.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 235-nm detector and a 4.6-mm ×25-cm column that contains 5-µm packing L1.The flow rate is about 1.0mLper minute.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the column efficiency is not less than 4000theoretical plates;the tailing factor is not more than 1.5;and the relative standard deviation for replicate injections is not more than 1.0%.
Procedure—
Separately inject equal volumes (about 25µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in mg,of C17H18N2O6in the portion of Nifedipine taken by the formula:
250C(rU/rS),
in which Cis the concentration,in mg per mL,of USP Nifedipine RSin the Standard preparation;and rUand rSare the peak responses obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Andrzej Wilk,Ph.D.,Senior Scientific Associate
Expert Committee:(PA5)Pharmaceutical Analysis 5
USP28–NF23Page 1375
Pharmacopeial Forum:Volume No.27(3)Page 2569
Phone Number:1-301-816-8305