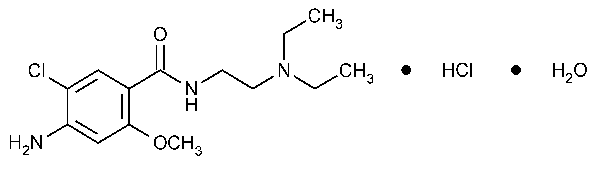
Metoclopramide Hydrochloride
Benzamide,4-amino-5-chloro-N-[2-(diethylamino)ethyl]-2-methoxy-,monohydrochloride,monohydrate.
4-Amino-5-chloro-N-[2-(diethylamino)ethyl]-o-anisamide monohydrochloride monohydrate [54143-57-6].
»Metoclopramide Hydrochloride contains not less than 98.0percent and not more than 101.0percent of C14H22ClN3O2·HCl,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight,light-resistant containers.
Identification—
A:
Infrared Absorption á197Mñ.
B:
Dissolve 50mg in 5mLof water,and add 5mLof a 1in 100solution of p-dimethylaminobenzaldehyde in 1Nhydrochloric acid:a yellow-orange color is produced.
C:
The RFvalue of the principal spot in the chromatogram of the Identification preparation corresponds to that of Standard preparation A,as obtained in the test for Chromatographic purity.
Water,Method Iá921ñ:
between 4.5%and 6.0%.
Residue on ignition á281ñ:
not more than 0.1%.
Chromatographic purity—
Standard preparations—
Dissolve USP Metoclopramide Hydrochloride RSin methanol,and mix to obtain a solution having a known concentration of 1mg per mL.Dilute quantitatively with methanol to obtain three Standard preparations,designated below by letters,having the following compositions:
| Standard
preparation |
Dilution | Concentration
(µg RS per mL) |
Percentage (%,
for comparison with test specimen) |
| A | (1in 4) | 250 | 0.5 |
| B | (3in 20) | 150 | 0.3 |
| C | (1in 20) | 50 | 0.1 |
Test preparation—
Dissolve an accurately weighed quantity of Metoclopramide Hydrochloride in methanol to obtain a solution containing 50mg per mL.
Identification preparation—
Dilute a portion of the Test preparationquantitatively with methanol to obtain a solution containing 500µg per mL.
Procedure—
Apply separately 10µLof the Test preparation,10µLof the Identification preparation,and 10µLof each Standard preparationto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel mixture.Allow the spots to dry,position the plate in a chromatographic chamber,and develop the chromatograms in a solvent system consisting of a mixture of chloroform,methanol,toluene,and ammonium hydroxide (140:60:20:1)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the developing chamber,mark the solvent front,and allow the solvent to evaporate.Examine the plate under short-wavelength UVlight,and compare the intensities of any secondary spots observed in the chromatogram of the Test preparationwith those of the principal spots in the chromatograms of the Standard preparations.[NOTE—Disregard any spots observed at the origins of the chromatograms.]No secondary spot from the chromatogram of the Test preparationis larger or more intense than the principal spot obtained from Standard preparation A(0.5%),and the sum of the intensities of all secondary spots obtained from the Test preparationcorresponds to not more than 1.0%.
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Assay—
Transfer about 300mg of Metoclopramide Hydrochloride,accurately weighed,to a stoppered,125-mLflask,add 10mLof mercuric acetate TSand 2mLof acetic anhydride,and allow to stand for 3hours.Add 80mLof glacial acetic acid,and titrate with 0.1Nperchloric acid VS,determining the endpoint potentiometrically (see Titrimetry á541ñ).Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 33.63mg of C14H22ClN3O2·HCl.
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(PA4)Pharmaceutical Analysis 4
USP28–NF23Page 1275
Pharmacopeial Forum:Volume No.29(5)Page 1536
Phone Number:1-301-816-8251