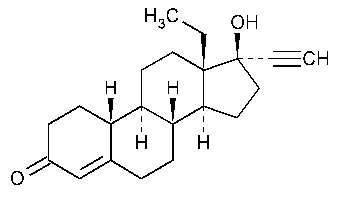
Levonorgestrel
18,19-Dinorpregn-4-en-20-yn-3-one,13-ethyl-17-hydroxy-,(17a)-(-)-.
(-)-13-Ethyl-17-hydroxy-18,19-dinor-17a-pregn-4-en-20-yn-3-one [797-63-7].
»Levonorgestrel contains not less than 98.0percent and not more than 102.0percent of C21H28O2,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed,light-resistant containers.
Identification—
A:
Infrared Absorption á197Kñ.
B:
Meeting the requirements of the tests for Specific rotationand Melting rangeprovides identification distinguishing it from norgestrel.
Melting range á741ñ:
between 232 and 239
and 239 ,but the range between beginning and end of melting does not exceed 4
,but the range between beginning and end of melting does not exceed 4 .
.
Specific rotation á781Sñ:
between -30 and -35
and -35 .
.
Test solution:
20mg per mL,in chloroform.
Loss on drying á731ñ—
Dry it at 105 for 5hours:it loses not more than 0.5%of its weight.
for 5hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.3%.
Limit of ethynyl group—
Dissolve 200mg in about 40mLof tetrahydrofuran.Add 10mLof silver nitrate solution (1in 10),and titrate with 0.1Nsodium hydroxide VS,using either a glass-calomel or a silver-silver chloride electrode system with potassium nitrate filling solution.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nsodium hydroxide is equivalent to 2.503mg of ethynyl group (-CºCH).Not less than 7.81%and not more than 8.18%of ethynyl group is found.
Chromatographic purity—
Proceed as directed in the test for Chromatographic purityunder Norgestrel.The requirements of the test are met if the sum of the impurities in the Test preparationdoes not exceed 2.0%and no single impurity is greater than 0.5%.
Assay—
Using USP Norgestrel RS,proceed with Levonorgestrel as directed in the Assayunder Norgestrel,except to read “Levonorgestrel”in place of “Norgestrel.”
Auxiliary Information—
Staff Liaison:Clydewyn M.Anthony,Ph.D.,Scientist
Expert Committee:(PA1)Pharmaceutical Analysis 1
USP28–NF23Page 1124
Pharmacopeial Forum:Volume No.28(4)Page 1245
Phone Number:1-301-816-8139