Iobenguane I131Injection
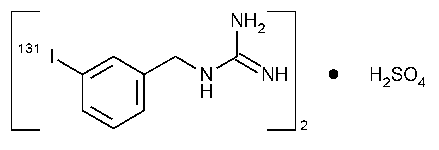
[[3-(Iodo-131I)-phenyl]methyl]guanidine sulfate (2:1).
[(m-Iodo-131I)-benzyl]guanidine sulfate (2:1).
»Iobenguane I131Injection is a sterile solution containing iobenguane sulfate in which a portion of the molecules contain radioactive iodine (131I)in the molecular structure.Iobenguane I131Injection contains not less than 90.0percent and not more than 110.0percent of the labeled amount of 131Ias iobenguane sulfate expressed in megabecquerels (or in millicuries)per mLat the time indicated in the labeling.It may contain preservatives or stabilizers.
Packaging and storage—
Preserve in single-dose or in multiple-dose containers that are adequately shielded.Store in a freezer.
Labeling—
Label it to include the following:the date of calibration;the amount of 131Ias iobenguane sulfate expressed as total megabecquerels (or millicuries)per mLat the time of calibration;the name and quantity of any added preservative or stabilizer;the expiration date;and the statement,“Caution—Radioactive Material.”The labeling indicates that,in making dosage calculations,correction is to be made for radioactive decay,and also indicates that the radioactive half-life of 131Iis 8.04days.
Radionuclidic identification (see Radioactivity á821ñ)—
Its gamma-ray spectrum is identical to that of a specimen of 131Iof known purity that exhibits a major photopeak having an energy of 0.364MeV.
Bacterial endotoxins á85ñ—
It contains not more than 175/VUSP Endotoxin Unit per mLof the Injection,when compared with USP Endotoxin RS,in which Vis the maximum recommended total dose,in mL,at the expiration date or time.
pHá791ñ:
between 4.5and 7.5.
Radionuclidic purity—
Using a suitable counting assembly (see Selection of a Counting Assemblyunder Radioactivity á821ñ),determine the radionuclidic purity of the Injection:not less than 99%of the total radioactivity is present as 131I.
Radiochemical purity—
Mobile phase—
Prepare a filtered and degassed mixture of water and acetonitrile (900:100).Add 3.04g of triethylamine per liter,and adjust with phosphoric acid to a pHof 4.0±0.4.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of iobenguane sulfate in water to obtain a solution having a known concentration of about 1mg per mL.
Test preparation—
Use Iobenguane I131Injection.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 229-nm detector,a collimated radiation detector (see Radioactivity á821ñ),and a 3.9-mm ×3-cm column that contains 10-µm packing L1.The flow rate is about 1.5mLper minute.Chromatograph the Standard preparation,and record the peak responses as directed under Procedure:the column efficiency is not less than 1000theoretical plates,the tailing factor is not more than 1.2,and the relative standard deviation for replicate injections is not more than 1.5%.
Procedure—
Inject a volume (about 20µL)of Injection,equivalent to between 1.6and 2.7MBq (or between 44and 64µCi)into the chromatograph,record the chromatograms,and measure the areas for the major peaks.The radioactivity of the iobenguane 131Ipeak is not less than 90%of the total radioactivity measured,and its retention time is within 10%of that of the Standard preparation,similarly chromatographed.The retention time of the iobenguane 131Ipeak obtained from the Injection is also within 10%of that obtained from the Standard preparation.
Other requirements—
It meets the requirements under Injections á1ñ,except that the Injection may be distributed or dispensed prior to the completion of the test for Sterility á71ñ,the latter test being started on the day of final manufacture,and except that it is not subject to the recommendation for Volume in Container.
Assay for radioactivity—
Using a counting assembly (see Selection of a Counting Assemblyunder Radioactivity á821ñ),determine the radioactivity,in MBq (or µCi),per mL,of Injection by use of a calibrated system as directed under Radioactivity á821ñ.
Auxiliary Information—
Staff Liaison:Andrzej Wilk,Ph.D.,Senior Scientific Associate
Expert Committee:(RMI)Radiopharmaceuticals and Medical Imaging Agents
USP28–NF23Page 1031
Phone Number:1-301-816-8305