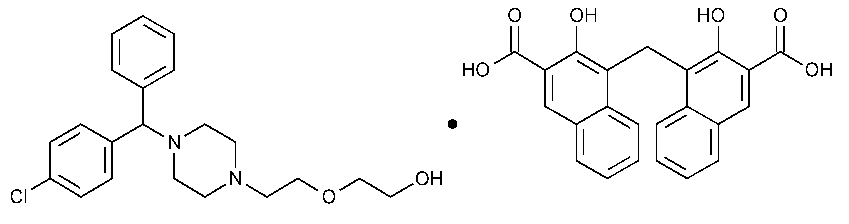
Hydroxyzine Pamoate
Ethanol,2-[2-[4-[(4-chlorophenyl)phenylmethyl]-1-piperazinyl]ethoxy]-,(±)-,compd.with 4,4¢-methylenebis[3-hydroxy-2-naphthalenecarboxylic acid](1:1).
(±)-2-[2-[4-(p-Chloro-a-phenylbenzyl)-1-piperazinyl]ethoxy]ethanol 4,4¢-methylenebis[3-hydroxy-2-naphthoate](1:1) [10246-75-0].
»Hydroxyzine Pamoate contains not less than 97.0percent and not more than 102.0percent of C21H27ClN2O2·C23H16O6,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
USP Reference standards á11ñ—
USP Hydroxyzine Pamoate RS.USP Hydroxyzine Hydrochloride RS.USP Pamoic Acid RS.
Identification—
A:
Transfer about 85mg (equivalent to about 50mg of hydroxyzine hydrochloride)to a separator containing 25mLof 1Nsodium hydroxide,and extract with three 20-mLportions of chloroform.Evaporate the combined chloroform extracts on a steam bath with the aid of a current of air to dryness,and dissolve the residue in 0.1Nhydrochloric acid to make 100mL.Dilute a 1-mLportion of this solution with 0.1Nhydrochloric acid to 50mL:the UVabsorption spectrum of the solution so obtained exhibits maxima and minima at the same wavelengths as that of a 1in 100,000solution of USP Hydroxyzine Hydrochloride RSin 0.1Nhydrochloric acid,concomitantly measured.
B:
Prepare a solution of it in a mixture of equal volumes of 0.1Nsodium hydroxide and acetone containing 2mg per mL.Apply 10µLof this solution and 10µLof a solution of USP Hydroxyzine Pamoate RSin the same medium,having a concentration of 2mg per mLto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.50-mm layer of chromatographic silica gel.Allow the spots to dry,and develop the chromatogram in 0.1Nhydrochloric acid until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the developing chamber,mark the solvent front,and locate the spots on the plate by viewing under long-wavelength UVlight:the RFvalue of the principal spot,representing the pamoate moiety,obtained from the test solution corresponds to that obtained from the Standard solution.Spray the plate lightly with potassium iodoplatinate TS:the RFvalue of the principal spot,representing the hydroxyzine moiety,obtained from the test solution corresponds to that obtained from the Standard solution.
Water,Method Iá921ñ:
not more than 5.0%.
Residue on ignition á281ñ:
not more than 0.5%.
Heavy metals,Method IIá231ñ:
0.005%.
Organic volatile impurities,Method Vá467ñ:
meets the requirements.
Solvent—
Use dimethyl sulfoxide.
Pamoic acid content—
Dissolve an accurately weighed quantity of USP Pamoic Acid RSin dimethylformamide to obtain a solution having a known concentration of about 0.45mg per mL.Transfer 2.0mLof the resulting solution to a 50-mLvolumetric flask,dilute with Mobile phase,prepared as directed in the Assay,to volume,and mix.Filter through a membrane filter of 0.5µm or finer porosity to obtain the Standard preparation.Chromatograph this Standard preparationas directed in the Assay.From the chromatogram of the Assay preparationobtained in the Assay,calculate the quantity,in mg,of pamoic acid (C23H16O6)in the portion of Hydroxyzine Pamoate taken by the formula:
2500C(rU/rS),
in which Cis the concentration,in mg per mL,of USP Pamoic Acid RSin the Standard preparation,and rUand rSare the pamoic acid peak responses obtained from the Assay preparationand the Standard preparation,respectively:the content of pamoic acid is between 49.4%and 51.9%,calculated on the anhydrous basis.
Assay—
Mobile phase—
Dissolve 8.65g of sodium 1-octanesulfonate in about 1000mLof water in a 2000-mLvolumetric flask,add 4.0mLof phosphoric acid,dilute with water to volume,mix,and filter through a membrane filter of 0.5µm or finer porosity.Prepare a suitable mixture of this solution and acetonitrile (45:55),making any adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Hydroxyzine Hydrochloride RSin dimethylformamide to obtain a solution having a known concentration of about 1mg per mL.Transfer 2.0mLof the resulting solution to a 100-mLvolumetric flask,dilute with Mobile phaseto volume,mix,and filter through a membrane filter of 0.5µm or finer porosity.
Assay preparation—
Transfer about 90mg of Hydroxyzine Pamoate,accurately weighed,to a 50-mLvolumetric flask,dissolve in dimethylformamide,dilute with dimethylformamide to volume,and mix.Transfer 2.0mLof the resulting solution to a 100-mLvolumetric flask,dilute with Mobile phaseto volume,mix,and filter through a membrane filter of 0.5µm or finer porosity,discarding the first 5mLof the filtrate.
Chromatographic system(seeChromatography á621ñ)—
The liquid chromatograph is equipped with a 230-nm detector and a 3.9-mm ×30-cm column that contains packing L1.The flow rate is about 1.5mLper minute.Chromatograph the Standard preparation,and record the peak responses as directed under Procedure:the column efficiency,calculated from the analyte peak,is not less than 2000theoretical plates,and the relative standard deviation for replicate injections is not more than 2%.Chromatograph the Assay preparation,and record the peak responses as directed under Procedure:the resolution,R,between hydroxyzine and pamoic acid is not less than 1.5.
Procedure—
Separately inject equal volumes (about 50µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.The relative retention times are about 0.5for hydroxyzine and 1.0for pamoic acid.Calculate the quantity,in mg,of C21H27ClN2O2·C23H16O6in the portion of Hydroxyzine Pamoate taken by the formula:
2500(763.27/447.83)C(rU/rS),
in which 763.27and 447.83are the molecular weights of hydroxyzine pamoate and hydroxyzine hydrochloride,respectively,Cis the concentration,in mg per mL,of USP Hydroxyzine Hydrochloride RSin the Standard preparation,and rUand rSare the hydroxyzine peak responses obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Salvador C.Salado,M.S.,Scientist and Latin American Liaison
Expert Committee:(PA3)Pharmaceutical Analysis 3
USP28–NF23Page 983
Phone Number:1-301-816-8165