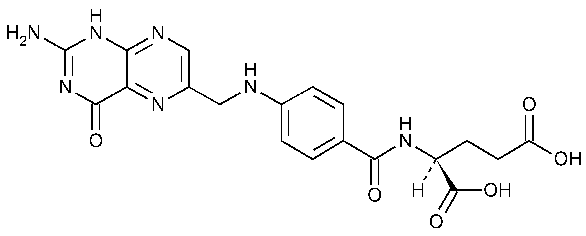
Folic Acid
L-Glutamic acid,N-[4-[[(2-amino-1,4-dihydro-4-oxo-6-pteridinyl)methyl]amino]benzoyl]-.
Folic acid.
N-[p-[[(2-Amino-4-hydroxy-6-pteridinyl)methyl]amino]-benzoyl]-L-glutamic acid [59-30-3].
»Folic Acid contains not less than 97.0percent and not more than 102.0percent of C19H19N7O6,calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed,light-resistant containers.
Identification,Ultraviolet Absorption á197Uñ—
Solution:
10µg per mL.
Medium:
Sodium hydroxide solution (1in 250).
Ratio:
A256/A365,between 2.80and 3.00.
Water—
Proceed as directed for Method Iá921ñ,except to stir the methanol solvent prior to and during the addition of the test specimen,and during the titration:not more than 8.5%is found.
Residue on ignition á281ñ:
not more than 0.3%.
Chromatographic purity—
3Nphosphoric acid
,6Nammonium hydroxide,Internal standard solution,Standard stock solution,Standard preparation,and Chromatographic system—Proceed as directed in the Assay.
Procedure—
Inject about 10µLof the Test solutioninto the chromatograph,and allow the Test solutionto elute for not less than 2times the retention time of folic acid.Record the chromatogram,and measure the responses of all the peaks.The sum of the area of all peaks,other than that due to folic acid,is not greater than 2.0%.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
[NOTE—Use low actinic glassware throughout the following procedure.]
3Nphosphoric acid—
Dissolve 9.8g of phosphoric acid in 100mLof water.
6Nammonium hydroxide—
Dilute 40mLof ammonium hydroxide with water to 100.0mL.
Mobile phase—
Transfer 2.0g of monobasic potassium phosphate to a 1000-mLvolumetric flask,and dissolve in about 650mLof water.Add 15.0mLof solution of 0.5Mtetrabutylammonium hydroxide in methanol,7.0mLof 3Nphosphoric acid,and 270mLof methanol.Cool to room temperature,and adjust with 3Nphosphoric acidor 6Nammonium hydroxideto a pHof 5.0,dilute with water to volume,mix,and filter.[NOTE—Recheck the pHbefore use.]
Internal standard solution—
Dissolve about 50mg of methylparaben in 1.0mLof methanol,dilute with Mobile phaseto 25.0mL,and mix.
Standard stock solution—
Prepare a solution of USP Folic Acid RSin Mobile phasehaving a known concentration of about 1mg per mL.[NOTE—Use 1mLof 10%ammonium hydroxide to dissolve the folic acid for every 100mLof Standard stock solutionprepared.]
Standard preparation—
Transfer 4.0mLof Standard stock solutionto a 50-mLvolumetric flask,add 4.0mLof Internal standard solution,dilute with Mobile phaseto volume,and mix.
Assay stock solution—
Transfer an accurately weighed quantity of about 100mg of Folic Acid to a 100-mLvolumetric flask,add about 40mLof Mobile phaseand 1mLof 10%ammonium hydroxide to dissolve.Dilute with Mobile phaseto volume,and mix.
Assay preparation—
Transfer 4.0mLof the Assay stock solutionto a 50-mLvolumetric flask,add 4.0mLof Internal standard solution,dilute with Mobile phaseto volume,and mix.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 280-nm detector and a 4.0-mm ×25-cm column that contains packing L1.The flow rate is about 1.2mLper minute.Chromatograph the Standard preparation,and record the peak responses as directed for Procedure:the resolution,R,between methylparaben and folic acid is not less than 3.6,and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in mg,of C19H19N7O6in the portion of Folic Acid taken by the formula:
1250C(RU/RS),
in which Cis the concentration,in mg per mL,of USP Folic Acid RS,on the anhydrous basis,in the Standard preparation;and RUand RSare the ratios of the response of the folic acid peak to that of the methylparaben peak obtained from the Assay preparationand Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Lawrence Evans,III,Ph.D.,Scientist
Expert Committee:(DSN)Dietary Supplements:Non-Botanicals
USP28–NF23Page 869
Pharmacopeial Forum:Volume No.29(2)Page 409
Phone Number:1-301-816-8389