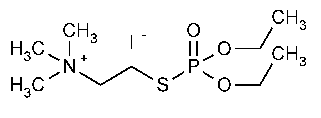
Echothiophate Iodide
Ethanaminium,2-[(diethoxyphosphinyl)thio]-N,N,N-trimethyl-,iodide.
(2-Mercaptoethyl)trimethylammonium iodide S-ester with O,O-diethyl phosphorothioate [513-10-0].
»Echothiophate Iodide contains not less than 95.0percent and not more than 100.5percent of C9H23INO3PS,calculated on the dried basis.
Packaging and storage—
Preserve in tight,light-resistant containers,preferably at a temperature below 0 .
.
Identification—
A:
Dissolve about 100mg in 2mLof water in a test tube,and add 1mLof nitric acid:a brown precipitate of iodine is formed.Transfer 1drop of this mixture to another test tube,add 1mLof solvent hexane,and shake:the solvent hexane takes on a pink color.
B:
Heat the remainder of the brown reaction mixture from Identificationtest Aover a flame until a colorless solution remains (about 3minutes).Cool,and dilute with water to about 10mL:a 2-mLportion of this solution responds to the tests for Phosphate á191ñ.
C:
A2-mLportion of the solution obtained in Identificationtest Bresponds to the tests for Sulfate á191ñ.
Loss on drying á731ñ—
Dry it in vacuum over phosphorus pentoxide at 50 for 3hours:it loses not more than 1.0%of its weight.
for 3hours:it loses not more than 1.0%of its weight.
Assay—
[NOTE—In the preparation of all reagents,and throughout this procedure,wherever water is specified,use only water that has been distilled,boiled for 10minutes,and cooled while protected from the atmosphere.]
pH12phosphate buffer—
Transfer 5.44g of anhydrous dibasic sodium phosphate to a 100-mLvolumetric flask,and add a volume of 1Nsodium hydroxide VSthat contains 36.5mEq of sodium hydroxide.Add about 40mLof water,shake to dissolve the sodium phosphate,dilute with water to volume,and mix.
0.004N Iodine—
Dilute 0.1Niodine with water to 0.004N,and standardize the solution on the day of use as follows.Weigh accurately about 150mg of arsenic trioxide,and dissolve in 20mLof 1Nsodium hydroxide,by warming if necessary,in a 500-mLvolumetric flask.Dilute with 40mLof water,add 2drops of methyl orange TS,then add 3Nhydrochloric acid until the yellow color is changed to pink.Add 2g of sodium bicarbonate,add water to volume,and mix.Transfer 5.0mLof this solution to a titration vessel,and add 50mLof pH7phosphate buffer (see under Solutionsin the section Reagents,Indicators,and Solutions).Titrate with 0.004Niodine,determining the endpoint potentiometrically,using platinum and silver-silver chloride electrodes.Calculate the normality.Each 197.8µg of arsenic trioxide is equivalent to 1mLof 0.004Niodine.
Procedure—
Dissolve about 125mg of Echothiophate Iodide,accurately weighed,in about 50mLof water in a 100-mLvolumetric flask,add water to volume,and mix.Transfer 10.0mLof this solution to a titration vessel containing 30mLof water.Add 10.0mLof pH12phosphate buffer,mix,cover,and allow to stand for 20minutes at 25±3 .Add 2mLof glacial acetic acid,rapidly and with mixing.Titrate with 0.004Niodine,determining the endpoint potentiometrically,using platinum and silver-silver chloride electrodes.Correct for the amount of free thiol sulfur by repeating the procedure but adding the glacial acetic acid first,then the pH12phosphate buffer,and titrating immediately.Subtract the volume of 0.004Niodineused in the second titration from that used in the first.Each mLof 0.004Niodineis equivalent to 1.533mg of C9H23INO3PS.
.Add 2mLof glacial acetic acid,rapidly and with mixing.Titrate with 0.004Niodine,determining the endpoint potentiometrically,using platinum and silver-silver chloride electrodes.Correct for the amount of free thiol sulfur by repeating the procedure but adding the glacial acetic acid first,then the pH12phosphate buffer,and titrating immediately.Subtract the volume of 0.004Niodineused in the second titration from that used in the first.Each mLof 0.004Niodineis equivalent to 1.533mg of C9H23INO3PS.
Auxiliary Information—
Staff Liaison:Lawrence Evans,III,Ph.D.,Scientist
Expert Committee:(PA6)Pharmaceutical Analysis 6
USP28–NF23Page 712
Phone Number:1-301-816-8389