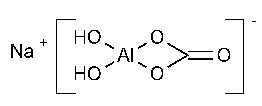
Dihydroxyaluminum Sodium Carbonate
Aluminum,[carbonato(1-)-O]dihydroxy-,monosodium salt.
Sodium (T-4)-[carbonato(2-)-O,O¢]dihydroxyaluminate(1-).
Sodium (carbonato)dihydroxyaluminate(1-) [539-68-4;16482-55-6].
»Dihydroxyaluminum Sodium Carbonate contains not less than 98.3percent and not more than 107.9percent of dihydroxyaluminum sodium carbonate (CH2AlNaO5),calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
A1-g portion,treated with 20mLof 3Nhydrochloric acid,dissolves with effervescence,and the resulting solution responds to the tests for Aluminum á191ñand to the flame test for Sodium á191ñ.
pHá791ñ:
between 9.9and 10.2in a suspension (1in 25).
Acid-neutralizing capacity á301ñ:
not less than 75.0%of the expected mEq value,determined on about 425mg of the undried material,accurately weighed,and calculated in relation to the results of the Assay.Each mg of NaAl(OH)2CO3has an expected acid-neutralizing capacity of 0.0278mEq.
Loss on drying á731ñ—
Dry it at 130 to constant weight:it loses not more than 14.5%of its weight.
to constant weight:it loses not more than 14.5%of its weight.
Isopropyl alcohol—
Isopropyl alcohol-free dihydroxyaluminum sodium carbonate—
Use a portion of Dihydroxyaluminum Sodium Carbonate that has been previously tested as directed in this section and found to be free of isopropyl alcohol.
Sodium chloride solution—
Prepare a solution (1in 5)of sodium chloride in water.
Standard stock solution—
Transfer about 1g of isopropyl alcohol,accurately weighed,to a 50-mLvolumetric flask containing about 10mLof Sodium chloride solution,dilute with Sodium chloride solutionto volume,and mix.This Standard stock solutioncontains about 20mg of isopropyl alcohol per mL.
Standard solutions—
Transfer 2.0,4.0,5.0,and 6.0mLof the Standard stock solutionto four separate 100-mLvolumetric flasks,dilute with Sodium chloride solutionto volume,and mix.These solutions contain about 0.4,0.8,1.0,and 1.2mg of isopropyl alcohol per mL,respectively.
Headspace containers—
Use suitable 20-mLcontainers capable of being tightly closed with an inert septum and a metallic crimp cap.
Standard preparations—
To four separate 20-mLHeadspace containersadd 1.0g of Isopropyl alcohol-free dihydroxyaluminum sodium carbonate.To the containers add,respectively,10.0mLof the appropriate Standard solution.These containers contain about 4,8,10,and 12mg of isopropyl alcohol,respectively.[NOTE—Keep the containers cool until sealed.]Seal the containers,place in a water bath maintained at 70 ,and allow to stand for 1hour.
,and allow to stand for 1hour.
Test preparation—
Transfer 1.0g of the Dihydroxyaluminum Sodium Carbonate under test to a Headspace container,and add 10.0mLof Sodium chloride solution.[NOTE—Keep the container cool until sealed.]Seal the container,place in a water bath maintained at 70 ,and allow to stand for 1hour.
,and allow to stand for 1hour.
Chromatographic system
(see Chromatography á621ñ)—The gas chromatograph is equipped with a flame-ionization detector and a 0.9-m ×3-mm column packed with support S3.The injection port temperature is maintained at 200 ,the detector temperature is maintained at 250
,the detector temperature is maintained at 250 ,and the column temperature is maintained at 180
,and the column temperature is maintained at 180 .Inject 1mLof the gaseous phase of the Standard preparationcontaining 10mg per container as directed for Procedure:the relative standard deviation for replicate injections is not more than 4%.
.Inject 1mLof the gaseous phase of the Standard preparationcontaining 10mg per container as directed for Procedure:the relative standard deviation for replicate injections is not more than 4%.
Procedure—
[NOTE—Use peak areas where peak responses are indicated.]Using a gas-tight syringe,separately inject equal volumes (about 1mL)of the gaseous headspace of the Standard preparationsand the Test preparationinto the gas chromatograph.Record the chromatograms,and measure the peak responses.Determine,based on a retention time comparison,if isopropyl alcohol is detected in the Test preparation.Plot the responses of the Standard preparationsversus the content,in mg,of isopropyl alcohol in each container,draw the straight line best fitting the plotted points,and calculate the correlation coefficient for the line.Asuitable system is one that yields a line having a correlation coefficient of not less than 0.99.From the graph,determine the total amount,TU,in mg,of isopropyl alcohol in the Test preparation.Calculate the percentage of isopropyl alcohol in the Dihydroxyaluminum Sodium Carbonate taken by the formula:
0.1TU/WU,
in which WUis the weight,in g,of the Dihydroxyaluminum Sodium Carbonate taken.The limit is 1.0%.
Sodium content—
Potassium chloride solution—
Prepare a solution of potassium chloride in water containing 38mg per mL.
Sodium chloride stock solution—
Dissolve a suitable quantity of sodium chloride,previously dried at 105 for 2hours and accurately weighed,in water,and dilute quantitatively and stepwise with water to obtain a solution containing 25.42µg per mL(10.0µg of sodium per mL).
for 2hours and accurately weighed,in water,and dilute quantitatively and stepwise with water to obtain a solution containing 25.42µg per mL(10.0µg of sodium per mL).
Standard solutions—
On the day of use,transfer 4.0mLof 1Nhydrochloric acid and 10.0mLof Potassium chloride solutionto each of two 100-mLvolumetric flasks.To the respective flasks add 5.0and 10.0mLof Sodium chloride stock solution.Dilute with water to volume,and mix.These solutions contain about 0.5and 1.0µg of sodium per mL,respectively.
Test solution—
Transfer about 250mg of Dihydroxyaluminum Sodium Carbonate,previously dried and accurately weighed,to a 200-mLvolumetric flask,add 40mLof 1Nhydrochloric acid,and boil for 1minute.Cool,dilute with water to volume,and mix.Transfer 10.0mLof this solution to a 100-mLvolumetric flask,dilute with water to volume,and mix.Transfer 5.0mLof this solution to a 100-mLvolumetric flask containing 4.0mLof 1Nhydrochloric acid and 10.0mLof Potassium chloride solution,dilute with water to volume,and mix.
Procedure—
Concomitantly determine the absorbances of the Standard solutionsand the Test solutionat the sodium emission line at 589.0nm with a suitable atomic absorption spectrophotometer (see Spectrophotometry and Light-scattering á851ñ)equipped with a sodium hollow-cathode lamp and an air–acetylene flame,using as a blank a solution prepared by pipeting 4mLof 1Nhydrochloric acid and 10.0mLof Potassium chloride solutioninto a 100-mLvolumetric flask,diluting with water to volume,and mixing.Plot the absorbances of the Standard solutionsversus concentrations,in µg per mLof sodium,and draw a straight line between the plotted points.From the graph so obtained,determine the concentration,C,in µg per mLof sodium,in the Test solution.Calculate the percentage of sodium in the portion of Dihydroxyaluminum Sodium Carbonate taken by the formula:
4000C/W,
in which Wis the quantity,in mg,of Dihydroxyaluminum Sodium Carbonate taken:between 15.2%and 16.8%is found.
Mercury,Method IIa á261ñ—
Transfer 2.0g to a 100-mLbeaker,and add 35mLof 1Nsulfuric acid:the limit is 1ppm.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Edetate disodium titrant—
Dissolve 18.6g of edetate disodium in water to make 500mL,and standardize as directed in the Assayunder Ammonium Alum.
Procedure—
Transfer about 300mg of undried Dihydroxyaluminum Sodium Carbonate,accurately weighed,to a 250-mLbeaker,add 10mLof 2Nsulfuric acid,cover the beaker,heat to 80 for 5minutes,and boil for 1minute.Add 30.0mLof 0.1Medetate disodium VS,again boil for 1minute,cool,and then add 10mLof acetic acid–ammonium acetate buffer TS,50mLof acetone,and 2mLof dithizone TS.Using a pHmeter,adjust with the addition of ammonium hydroxide or dilute sulfuric acid to a pHof 4.5,and titrate with 0.05Mzinc sulfate VS,maintaining the pHat 4.5by the addition of ammonium hydroxide as necessary,to an orange-pink color.Perform a blank determination,and make any necessary correction.Each mLof 0.1M Edetate disodium titrantis equivalent to 14.40mg of CH2AlNaO5.
for 5minutes,and boil for 1minute.Add 30.0mLof 0.1Medetate disodium VS,again boil for 1minute,cool,and then add 10mLof acetic acid–ammonium acetate buffer TS,50mLof acetone,and 2mLof dithizone TS.Using a pHmeter,adjust with the addition of ammonium hydroxide or dilute sulfuric acid to a pHof 4.5,and titrate with 0.05Mzinc sulfate VS,maintaining the pHat 4.5by the addition of ammonium hydroxide as necessary,to an orange-pink color.Perform a blank determination,and make any necessary correction.Each mLof 0.1M Edetate disodium titrantis equivalent to 14.40mg of CH2AlNaO5.
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(PA4)Pharmaceutical Analysis 4
USP28–NF23Page 653
Phone Number:1-301-816-8251