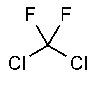
Dichlorodifluoromethane
»Dichlorodifluoromethane contains not less than 99.6percent and not more than 100.0percent of CCl2F2,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight cylinders,and avoid exposure to excessive heat.
Identification—
The IRabsorption spectrum of it,determined in a 10-cm cell with sodium chloride windows,at atmospheric pressure,exhibits maxima,among others,at the following wavelengths,in µm:4.33(m),4.46(m),4.56(m),6.29(m),7.25(m),8.05(s),8.63(s),9.1(vs),10.7(vs),10.8(vs),11.2(m),11.3(m),13.1(w),and 13.9(w).The stronger maxima are best obtained at pressures less than 10mm of mercury.
Boiling temperature:
approximately -30 ,determined as directed under Approximate Boiling Temperature(see Aerosols,Nasal Sprays,Metered-Dose Inhalers,and Dry Powder Inhalers á601ñ).
,determined as directed under Approximate Boiling Temperature(see Aerosols,Nasal Sprays,Metered-Dose Inhalers,and Dry Powder Inhalers á601ñ).
Water:
not more than 0.001%,determined as directed under Water Content(see Aerosols,Nasal Sprays,Metered-Dose Inhalers,and Dry Powder Inhalers á601ñand Method Icunder Water Determination á921ñ).
High-boiling residues:
not more than 0.01%,determined as directed for High-Boiling Residues,Method I,under Aerosols,Nasal Sprays,Metered-Dose Inhalers,and Dry Powder Inhalers á601ñ.
Chromatographic purity—
In the chromatogram recorded for the Assay,identify the peaks due to dichlorotetrafluoroethane and trichloromonofluoromethane from relative retention times observed on chromatographing the Resolution preparation:the sum of these two peak areas is not greater than 0.2%of the total of all peak areas,and the sum of the areas of all peaks other than that for dichlorodifluoromethane is not greater than 0.4%of the total of all peak areas.
Inorganic chlorides—
Place 5mLof anhydrous methanol in a test tube,add 3drops of a saturated solution of silver nitrate in anhydrous methanol,shake,and add 7g of Dichlorodifluoromethane:no opalescence or turbidity is produced.
Assay—
Resolution preparation—
Introduce a liquid-phase mixture of dichlorodifluoromethane,dichlorotetrafluoroethane,and trichloromonofluoromethane into an evacuated headspace vial.
Assay preparation—
Introduce the liquid phase of Dichlorodifluoromethane into an evacuated headspace vial.
Chromatographic system
(see Chromatography á621ñ)—The gas chromatograph is equipped with a flame-ionization detector,a headspace sampler,and a 2-mm ×1.8-m stainless steel column containing 1%phase G25on support S12.For the headspace sampler,the bath temperature is 100 ,the valve/loop temperature is 105
,the valve/loop temperature is 105 ,and the sampling time is 3seconds.[NOTE—Make adjustments as necessary to optimize peak areas to record trace level impurities.]The carrier gas is helium maintained at a flow rate of 20mLper minute.The injection port and detector temperatures are maintained at 110
,and the sampling time is 3seconds.[NOTE—Make adjustments as necessary to optimize peak areas to record trace level impurities.]The carrier gas is helium maintained at a flow rate of 20mLper minute.The injection port and detector temperatures are maintained at 110 and 200
and 200 ,respectively.The column temperature is programmed to rise from 70
,respectively.The column temperature is programmed to rise from 70 at the time of injection to 170
at the time of injection to 170 at the rate of 10
at the rate of 10 per minute,after which it is maintained at 170
per minute,after which it is maintained at 170 for 5minutes.Inject the gas phase headspace Resolution preparationand obtain the chromatogram as directed for Procedure:the approximate relative retention times are 0.5for dichlorodifluoromethane,0.8for dichlorotetrafluoroethane,and 1.0for trichloromonofluoromethane;and the resolution,R,between dichlorotetrafluoroethane and trichloromonofluoromethane is not less than 2.0.
for 5minutes.Inject the gas phase headspace Resolution preparationand obtain the chromatogram as directed for Procedure:the approximate relative retention times are 0.5for dichlorodifluoromethane,0.8for dichlorotetrafluoroethane,and 1.0for trichloromonofluoromethane;and the resolution,R,between dichlorotetrafluoroethane and trichloromonofluoromethane is not less than 2.0.
Procedure—
Inject the gas phase headspace Assay preparationinto the chromatograph,record the chromatogram,and measure the peak areas.Calculate the percentage (a/a)of CCl2F2in the portion of Dichlorodifluoromethane taken.
Auxiliary Information—
Staff Liaison:Karen A Russo,Ph.D.,Scientist
Expert Committee:(PA1)Pharmaceutical Analysis 1
USP28–NF23Page 2997
Pharmacopeial Forum:Volume No.27(1)Page 1825
Phone Number:1-301-816-8379