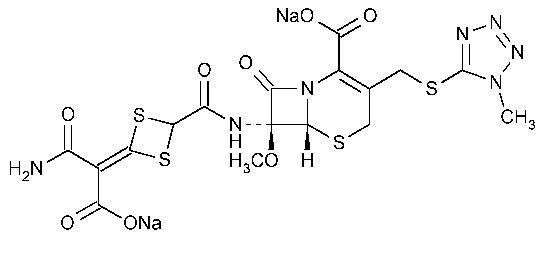
Cefotetan Disodium
5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,7-[[[4-[[(2-amino-1-carboxy-2-oxoethylidene)-1,3-dithietan-2-yl]carbonyl]amino]-7-methoxy-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-,disodium salt,[6R-(6a,7a)]-.
(6R,7S)-4-[[2-Carboxy-7-methoxy-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-7-yl]carbamoyl]-1,3-dithietane-D2,a-malonamic acid,disodium salt.
(6R,7S)-7-[4-(Carbamoylcarboxymethylene)-1,3-dithietane-2-carboxamido]-7-methoxy-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,disodium salt [74356-00-6].
»Cefotetan Disodium contains the equivalent of not less than 830µg and not more than 970µg of cefotetan (C17H17N7O8S4)per mg,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
Labeling—
Where it is intended for use in preparing injectable dosage forms,the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
Identification—
A:
The retention time of the major peak in the chromatogram of the Assay preparationcorresponds to that of the Standard preparationobtained as directed in the Assay.
B:
It responds to the tests for Sodium á191ñ.
pHá791ñ:
between 4.0and 6.5,in a solution (1in 10).
Water,Method ICá921ñ:
not more than 1.5%.
Other requirements—
Where the label states that Cefotetan Disodium is sterile,it meets the requirements for Sterilityand Bacterial endotoxinsunder Cefotetan for Injection.Where the label states that Cefotetan Disodium must be subjected to further processing during the preparation of injectable dosage forms,it meets the requirements for Bacterial endotoxinsunder Cefotetan for Injection.
Assay—
[NOTE—Protect the Standard preparation,the Resolution solution,and the Assay preparations from light,and use within 2hours.]
Mobile phase,Standard preparation,Resolution solution,and Chromatographic system—
Proceed as directed in the Assayunder Cefotetan.
Assay preparation—
Transfer about 40mg of Cefotetan Disodium,accurately weighed,to a 200-mLvolumetric flask,add 10mLof methanol,swirl for several minutes,add 10mLof acetonitrile,and swirl until dissolved.Dilute with water to volume,and mix.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 392
Phone Number:1-301-816-8335