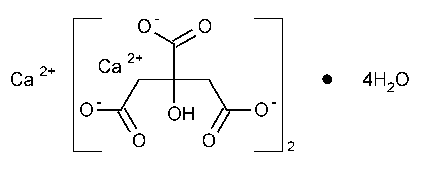
Calcium Citrate
1,2,3-Propanetricarboxylic acid,2-hydroxy-,calcium salt (2:3),tetrahydrate.
Calcium citrate (3:2),tetrahydrate [5785-44-4].
»Calcium Citrate contains four molecules of water of hydration.When dried at 150 to constant weight,it contains not less than 97.5percent and not more than 100.5percent of Ca3(C6H5O7)2.
to constant weight,it contains not less than 97.5percent and not more than 100.5percent of Ca3(C6H5O7)2.
Packaging and storage—
Preserve in well-closed containers.
Identification—
A:
Dissolve 0.5g in a mixture of 10mLof water and 2.5mLof 2Nnitric acid.Add 1mLof mercuric sulfate TS,heat to boiling,and add 1mLof potassium permanganate TS:a white precipitate is formed.
B:
Ignite completely 0.5g at as low a temperature as possible,cool,and dissolve the residue in a mixture of 10mLof water and 1mLof glacial acetic acid.Filter,and add 10mLof ammonium oxalate TSto the filtrate:a voluminous white precipitate is formed,and it is soluble in hydrochloric acid.
Loss on drying á731ñ—
Dry it at 150 for 4hours:it loses between 10.0%and 13.3%of its weight.
for 4hours:it loses between 10.0%and 13.3%of its weight.
Arsenic,Method Iá211ñ—
Dissolve 1g in 5mLof 3Nhydrochloric acid,and dilute with water to 35mL:the limit is 3µg per g.
Limit of fluoride—
[NOTE—Prepare and store all solutions in plastic containers.]
Standard solution—
Dissolve an accurately weighed quantity of USP Sodium Fluoride RSquantitatively in water to obtain a solution containing 2.210mg per mL.On the day of use,transfer 5.0mLof this solution to a 1000-mLvolumetric flask,dilute with water to volume,and mix.Each mLof this solution contains 5µg of fluoride ion.
Electrode system—
Use a fluoride-specific,ion-indicating electrode and a silver–silver chloride reference electrode connected to a pHmeter capable of measuring potentials with a minimum reproducibility of ±0.2mV(see pHá791ñ).
Standard response line—
Transfer 1.0,5.0,and 10.0mLof the Standard solutionto separate 250-mLplastic beakers,to each add 50mLof water,5mLof 1Nhydrochloric acid,10mLof 1.0Msodium citrate,and 10mLof 0.2Medetate disodium,and mix.If necessary,adjust with 1Nsodium hydroxide or 1Nhydrochloric acid to a pHof 5.5.Transfer each solution to a separate 100-mLvolumetric flask,dilute with water to volume,and mix.Transfer 50mLof each solution to separate 250-mLplastic beakers,and measure the potential of each solution with the Electrode system.Between each reading wash the electrodes with water,and absorb any residual water by blotting the electrodes dry.Plot the logarithms of the fluoride concentrations (0.05,0.25,and 0.50µg per mL,respectively)versus potential,in mV.
Procedure—
Transfer 1.0g of Calcium Citrate to a 100-mLbeaker,add 10mLof water and,while stirring,10mLof 1Nhydrochloric acid.When dissolved,boil rapidly for 1minute,transfer the solution to a 250-mLplastic beaker,and cool in ice water.Add 15mLof 1.0Msodium citrate and 10mLof 0.2Medetate disodium,and adjust with 1Nsodium hydroxide or 1Nhydrochloric acid to a pHof 5.5.Transfer this solution to a 100-mLvolumetric flask,dilute with water to volume,and mix.Transfer 50mLof this solution to a 250-mLplastic beaker,and measure the potential of this test solution with the Electrode system.From the measured potential and the Standard response linedetermine the concentration,C,in µg per mL,of fluoride ion in the test solution.Calculate the percentage of fluoride in the specimen taken by multiplying Cby 0.01:the limit is 0.003%.
Limit of acid-insoluble substances—
Dissolve 5g by heating with a mixture of 10mLof hydrochloric acid and 50mLof water for 30minutes:the residue so obtained,filtered,washed,and dried at 105 for 2hours,weighs not more than 10mg (0.2%).
for 2hours,weighs not more than 10mg (0.2%).
Lead á251ñ—
Prepare a Test Preparationby dissolving 0.5g in 20mLof 3Nhydrochloric acid,evaporating on a steam bath to about 10mL,diluting with water to about 20mL,and cooling.Use 5mLof Diluted Standard Lead Solution(5µg of Pb)for the test:the limit is 0.001%.
Heavy metals,Method Iá231ñ—
Dissolve 1g in a mixture of 20mLof water and 2mLof hydrochloric acid,add 1.5mLof ammonium hydroxide,and dilute with water to 25mL:the limit is 0.002%.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Dissolve about 350mg of Calcium Citrate,previously dried at 150 to constant weight and accurately weighed,in a mixture of 10mLof water and 2mLof 3Nhydrochloric acid,and dilute with water to about 100mL.While stirring,add about 30mLof 0.05Medetate disodium VSfrom a 50-mLburet.Add 15mLof 1Nsodium hydroxide and 300mg of hydroxy naphthol blue,and continue the titration to a blue endpoint.Each mLof 0.05Medetate disodium is equivalent to 8.307mg of Ca3(C6H5O7)2.
to constant weight and accurately weighed,in a mixture of 10mLof water and 2mLof 3Nhydrochloric acid,and dilute with water to about 100mL.While stirring,add about 30mLof 0.05Medetate disodium VSfrom a 50-mLburet.Add 15mLof 1Nsodium hydroxide and 300mg of hydroxy naphthol blue,and continue the titration to a blue endpoint.Each mLof 0.05Medetate disodium is equivalent to 8.307mg of Ca3(C6H5O7)2.
Auxiliary Information—
Staff Liaison:Karen A Russo,Ph.D.,Scientist
Expert Committee:(PA1)Pharmaceutical Analysis 1
USP28–NF23Page 324
Pharmacopeial Forum:Volume No.27(6)Page 3257
Phone Number:1-301-816-8379