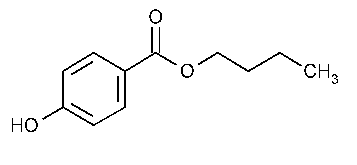
Butylparaben
»Butylparaben contains not less than 99.0percent and not more than 100.5percent of C11H14O3,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification,Infrared Absorption á197Mñ.
Acidity—
Heat 0.75g in 15mLof water at 80 for 1minute,cool,and filter:the filtrate is neutral or acid to litmus.To 10mLof the filtrate add 0.20mLof 0.10Nsodium hydroxide and 2drops of methyl red TS:the solution is yellow.
for 1minute,cool,and filter:the filtrate is neutral or acid to litmus.To 10mLof the filtrate add 0.20mLof 0.10Nsodium hydroxide and 2drops of methyl red TS:the solution is yellow.
Loss on drying á731ñ—
Dry it over silica gel for 5hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.05%.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Transfer about 2g of Butylparaben,accurately weighed,to a flask fitted with a ground-glass stopper and equipped for refluxing under a water-cooled condenser.Add 40.0mLof 1Nsodium hydroxide VS,and reflux for 1hour.Cool to room temperature,and rinse the condenser with water.Titrate the excess sodium hydroxide with 1Nsulfuric acid VS,continuing the titration until the second point of inflection and determining the endpoint potentiometically (see Titrimetry á541ñ).Perform a blank determination (see Residual Titrations underTitrimetry á541ñ).Each mLof 1Nsodium hydroxide is equivalent to 194.2mg of C11H14O3.
Auxiliary Information—
Staff Liaison:Justin Lane,B.S.,Scientific Associate
Expert Committee:(EMC)Excipients:Monograph Content
USP28–NF23Page 2969
Pharmacopeial Forum:Volume No.30(4)Page 1431
Phone Number:1-301-816-8323