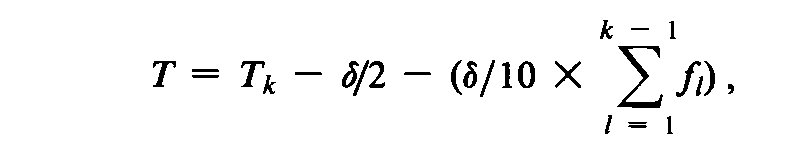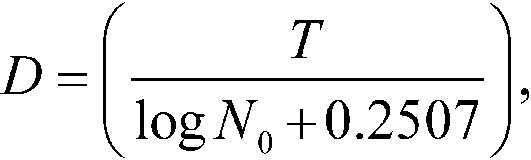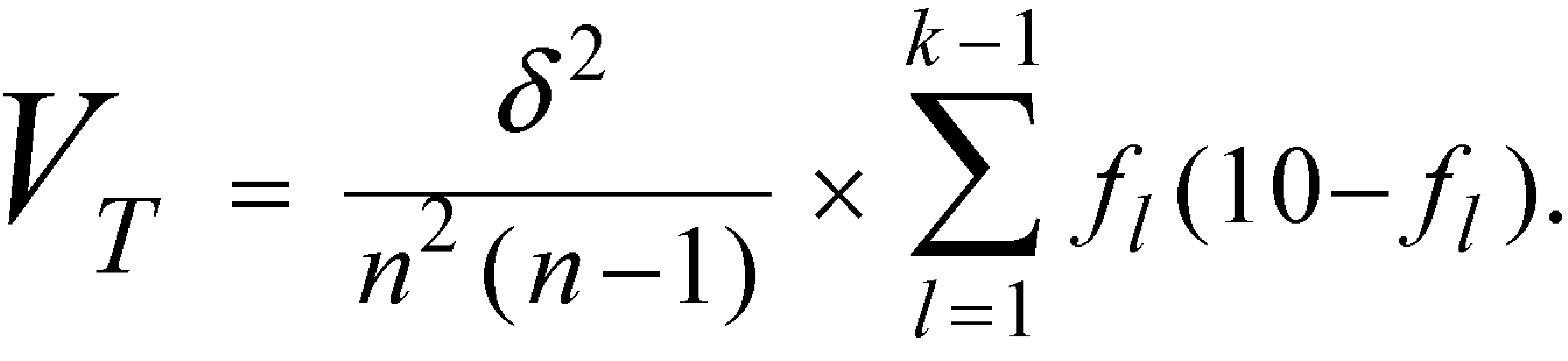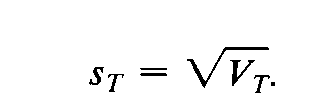á55ñBIOLOGICAL INDICATORS—RESISTANCE PERFORMANCE TESTS
TOTAL VIABLE SPORE COUNT
Remove three specimens of the relevant biological indicator from their original individual containers.Disperse the paper into component fibers by placing the test specimens in a sterile 250-mLcup of a suitable blender containing 100mLof chilled,sterilized Purified Water and blending for 3to 5minutes to achieve a homogeneous suspension.Transfer a 10-mLaliquot of the suspension to a sterile,screw-capped 16-×125-mm tube.For Biological Indicator for Steam Sterilization,Paper Carrier,heat the tube containing the suspension in a water bath at 95 to 100
to 100 for 15minutes (heat shock),starting the timing when the temperature reaches 95
for 15minutes (heat shock),starting the timing when the temperature reaches 95 .For Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and for Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,heat the tube containing the suspension in a water bath at 80
.For Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and for Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,heat the tube containing the suspension in a water bath at 80 to 85
to 85 for 10minutes,starting the timing when the temperature reaches 80
for 10minutes,starting the timing when the temperature reaches 80 .Cool rapidly in an ice water bath at 0
.Cool rapidly in an ice water bath at 0 to 4
to 4 .Transfer two 1-mLaliquots to suitable tubes,and make appropriate serial dilutions in sterilized Purified Water,the dilutions being selected as calculated to yield preferably 30to 300colonies,but not less than 6,on each of a pair of plates when treated as described below.Where the biological indicator has a low spore concentration,it may be necessary to modify the dilution series and to use more plates at each dilution.Prepare a separate series of plates for each aliquot.Place 1.0mLof each selected dilution in each of two 15-×100-mm Petri dishes.Within 20minutes,add to each plate 20mLof Soybean–Casein Digest Agar Medium(see Microbial Limit Tests á61ñ)that has been melted and cooled to 45
.Transfer two 1-mLaliquots to suitable tubes,and make appropriate serial dilutions in sterilized Purified Water,the dilutions being selected as calculated to yield preferably 30to 300colonies,but not less than 6,on each of a pair of plates when treated as described below.Where the biological indicator has a low spore concentration,it may be necessary to modify the dilution series and to use more plates at each dilution.Prepare a separate series of plates for each aliquot.Place 1.0mLof each selected dilution in each of two 15-×100-mm Petri dishes.Within 20minutes,add to each plate 20mLof Soybean–Casein Digest Agar Medium(see Microbial Limit Tests á61ñ)that has been melted and cooled to 45 to 50
to 50 .Swirl to attain a homogeneous suspension,and allow to solidify.Incubate the plates in an inverted position at 55
.Swirl to attain a homogeneous suspension,and allow to solidify.Incubate the plates in an inverted position at 55 to 60
to 60 for Biological Indicator for Steam Sterilization,Paper Carrier,and at 30
for Biological Indicator for Steam Sterilization,Paper Carrier,and at 30 to 35
to 35 for Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,and for Biological Indicator for Dry-Heat Sterilization,Paper Carrier,or at the optimal recovery temperature specified by the manufacturer,and examine the plates after 24and 48hours,recording for each plate the number of colonies,and using the number of colonies after 48hours to calculate the results.Calculate the average number of spores per specimen from the results,using the appropriate dilution factor.The test is valid if the log number of spores per Carrier at 48hours is equal to or greater than the log number after 24hours in each case.For Biological Indicator for Steam Sterilization,Self-Contained,aseptically remove the spore strip from the container,and proceed as directed for Biological Indicator for Steam Sterilization,Paper Carrier.
for Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,and for Biological Indicator for Dry-Heat Sterilization,Paper Carrier,or at the optimal recovery temperature specified by the manufacturer,and examine the plates after 24and 48hours,recording for each plate the number of colonies,and using the number of colonies after 48hours to calculate the results.Calculate the average number of spores per specimen from the results,using the appropriate dilution factor.The test is valid if the log number of spores per Carrier at 48hours is equal to or greater than the log number after 24hours in each case.For Biological Indicator for Steam Sterilization,Self-Contained,aseptically remove the spore strip from the container,and proceed as directed for Biological Indicator for Steam Sterilization,Paper Carrier.
D VALUE DETERMINATION
For all tests described in this section,handle each test specimen with aseptic precautions,using sterilized equipment where applicable.
Apparatus
For Biological Indicator for Dry-Heat Sterilization,Paper Carrier,use an apparatus of known thermodynamic characteristics that has been validated for compliance with the requirements for safety1and performance,2that consists of a sterilizing chamber equipped with a means of heating the contained air,preferably electrically rather than gas fired,and that has adequate movement of the air through forced ventilation (by mechanical devices such as blowers),with sensing and control devices for temperature and timing capable of indicating with an accuracy of not more than 0.5 and 1-second intervals,respectively.The geometrical pattern of the heat source(s)is such as to enable the biological indicators under test to be uniformly heated under the specified conditions.The temperature profile in the chamber is known,and cold spots,hot spots,and slow heat zones identified.The chamber has the capability to work within a temperature range of 40
and 1-second intervals,respectively.The geometrical pattern of the heat source(s)is such as to enable the biological indicators under test to be uniformly heated under the specified conditions.The temperature profile in the chamber is known,and cold spots,hot spots,and slow heat zones identified.The chamber has the capability to work within a temperature range of 40 to 300
to 300 ,with an accuracy at any particular setting of not less than ±2
,with an accuracy at any particular setting of not less than ±2 .The apparatus is equipped with a suitable additional access door or port so as to enable the entry and insertion (or removal)of specimens within 6seconds and to enable the temperature to return to the set temperature within 0.5minute where the specified temperature is 120
.The apparatus is equipped with a suitable additional access door or port so as to enable the entry and insertion (or removal)of specimens within 6seconds and to enable the temperature to return to the set temperature within 0.5minute where the specified temperature is 120 to 190
to 190 and within 1.0minute where such temperature is 220
and within 1.0minute where such temperature is 220 and above.
and above.
For Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,use an apparatus that consists of a test chamber with a means of ensuring adequate mixing of the sterilant gas and a means of heating the sterilant gas to not lower than the preselected operating temperature so that no liquid enters the test chamber,equipped with temperature control and monitoring,pressure control,humidification,and gas concentration monitoring devices.Detailed specifications and operational parameters for suitable apparatus are those published in Standard for a Biological Indicator—Evaluator Resistometer for Ethylene Oxide Gas Vessels(BIER/EO)Gas Vessels.3
For Biological Indicator for Steam Sterilization,Paper Carrier,and for Biological Indicator for Steam Sterilization,Self-Contained,use an apparatus that consists of a chamber equipped with heating,temperature,and steam control and monitoring devices.Detailed specifications and operational parameters for suitable apparatus are those published in Standard for a Biological Indicator—Evaluator Resistometer for Saturated Steam(BIER/Steam Vessels).4
Procedure
Carry out the tests for Dvalueat each of the applicable sets of sterilization conditions for which the packaged biological indicator under test is labeled for use.Take a sufficient number of groups of specimens of biological indicators in their original individual containers,each group consisting of 5to 10specimens.The number of groups provides a range of observations from not less than one labeled Dvaluebelow the labeled survival time through not less than one labeled Dvalueabove the labeled kill time.Place each group on a separate suitable specimen holder that permits each specimen to be exposed to the prescribed sterilizing condition at a specific location in the sterilizing chamber.Check the apparatus for operating parameters using specimen holders without specimens.Select a series of sterilizing times in increments from the shortest time for the specimens to be tested.The differences in sterilizing times over the series are as constant as feasible,and the difference between adjacent times is no greater than 75%of the labeled Dvalue.
For Biological Indicator for Dry-Heat Sterilization,Paper Carrier,preheat the sterilizing chamber for 30minutes.Open the access door or port,place one of the holders with a group of specimens in the sterilizing chamber,close the access door or port,and continue to operate the apparatus.Commence timing the heat exposure when the chamber temperature returns to 2 below the specified temperature.After the contents have been subjected to the sterilizing condition for a predetermined time selected from a series of time increments,remove the holder with the heated specimens,and replace it with another holder with specimens.Repeat the sterilizing procedure similarly,but for another predetermined time,and continue with successive groups until all have been heated appropriately.
below the specified temperature.After the contents have been subjected to the sterilizing condition for a predetermined time selected from a series of time increments,remove the holder with the heated specimens,and replace it with another holder with specimens.Repeat the sterilizing procedure similarly,but for another predetermined time,and continue with successive groups until all have been heated appropriately.
For Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,proceed as follows:
-
Evacuate the test chamber to a pressure of not more than 100±3mm of mercury.
-
Inject sufficient water vapor (e.g.,saturated steam)to bring the chamber contents to within 10%relative humidity of the required humidification condition,and allow the chamber to equilibrate with moisture and to temperature for about 30minutes.
-
Inject a sufficient quantity of temperature-equilibrated ethylene oxide gas to attain the appropriate concentration ±30mg of ethylene oxide per liter.
-
Subject a group of specimens to the appropriate temperature,humidification,and gas concentration conditions for the required time.
-
Evacuate the test chamber to a pressure of 100±3mm of mercury,and release the vacuum with sterile filtered air.Repeat this until not less than 99%of the remaining gas has been removed,and remove the holder(s)with the exposed specimens.For exposing further groups of specimens to the sterilization conditions,proceed with steps 6and 7.
-
Flush the test chamber five times with filtered air after evacuation each time to a pressure of not more than 100±3mm of mercury.
-
Repeat the entire sterilizing procedure,steps 1through 6,for other groups of unexposed specimens,but maintain the specified conditions of step 4for each of the other required times.
For Biological Indicator for Steam Sterilization,Paper Carrier,exhaust the sterilizing chamber,and within 15seconds of opening the door,place one of the holders with a group of specimens in the sterilizing chamber,and operate the apparatus to heat up the chamber contents as quickly as possible.After the contents have been subjected to the sterilizing condition for a predetermined time selected from the series of time increments,exhaust the chamber as quickly as possible.Remove the holder with the heated specimens,and replace it with another group of specimens.Repeat the sterilizing procedure similarly,but for another predetermined time,and continue with successive groups until all have been appropriately heated.
For Biological Indicator for Steam Sterilization,Self-Contained,follow the procedure indicated for Biological Indicator for Steam Sterilization,Paper Carrier,but handle each self-contained unit as a biological indicator system,with the Dvaluedetermined for the self-contained system.
Recovery
After completion of the sterilizing procedure for Biological Indicator for Dry-Heat Sterilization,Paper Carrier;Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier;or Biological Indicator for Steam Sterilization,Paper Carrier,whichever is applicable,and within a noted time not more than 4hours,aseptically remove and add each strip to 10to 30mLof Soybean–Casein Digest Medium(see Mediaunder Sterility Tests á71ñ)to submerge the biological indicator completely in a suitable tube.For each Biological Indicator for Steam Sterilization,Self-Containedspecimen,the paper strip is immersed in the self-contained medium according to manufacturers'instructions,within a noted time not more than 4hours.Incubate each tube at a temperature of 55 to 60
to 60 for Biological Indicator for Steam Sterilization,Paper Carrier,and Biological Indicator for Steam Sterilization,Self-Contained,or at 30
for Biological Indicator for Steam Sterilization,Paper Carrier,and Biological Indicator for Steam Sterilization,Self-Contained,or at 30 to 35
to 35 for Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,or in any case at the optimal recovery temperature specified by the manufacturer.Observe each inoculated medium-containing tube at 24and 48hours,and every 1or 2days thereafter for a total of 7days after inoculation.(Where growth is observed at any particular observation time,further incubation of the specimen(s)concerned may be omitted.)Note the number of specimens showing no evidence of growth at any time.
for Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,or in any case at the optimal recovery temperature specified by the manufacturer.Observe each inoculated medium-containing tube at 24and 48hours,and every 1or 2days thereafter for a total of 7days after inoculation.(Where growth is observed at any particular observation time,further incubation of the specimen(s)concerned may be omitted.)Note the number of specimens showing no evidence of growth at any time.
Calculation
This chapter describes the use of the Limited Spearman-Karber Method for determining the Dvalueof biological indicators on spore paper carriers.Use this method in the event of a compendial issue or regulatory referee testing of a biological indicator system.It is recognized that other methods,such as the Survival Curve Method and the Stumbo-Murphy-Cochran procedure,may be routinely used by manufacturers and users of biological indicators to determine Dvalues.The calculation of the Dvalue using the Limited Spearman-Karber Method is based on the use of 10biological indicators per group.[NOTE—If less than 10biological indicators are used (i.e.,5),the formula and the various calculation steps will have to be modified,including the Replacement of Missing Values;however,the requirements of the test remain the same.]
Designate the number of specimens taken for each group (i.e.,10)by n,and the difference between adjacent times (in minutes)by d.Designate for each group of the series the number of specimens showing no growth by:
f1,f2,...fk,
in which f1is the response of all 10specimens showing growth (0/10inactivated)in the group held for the shortest time for such result that is adjacent to an intermediate mortality;and fkis the response of all 10specimens of the group showing no growth (10/10inactivated)in the group held for the longest time for such result that is adjacent to an intermediate mortality.Do not use for the calculations observations for groups beyond the ends of the series,f1and fk,giving results that are not adjacent to an intermediate mortality.The test is valid if there is available a result (0/10)from a group held for a shorter time than that for the selected shortest time result (f1),and there is available a result (10/10)from a group held for a longer time than that for the selected longest time result (fk).Calculate the mean heating time,T,for achieving complete kill by the equation:
in which Tkis the time for achieving the result fk.Calculate the Dvalueby the equation:
in which N0is the average spore count per carrier determined by Total Viable Spore Count(see above)at the time of making this test.Calculate the variance of T,VT,by the equation:
in which drepresents a constant interval between successive exposures,as defined above.
The standard deviation,sT,is the square root of the variance:
Calculate the lower and upper 95%confidence limits (approximate CL)for the Dvalueby the equation:
approximate CLfor D=(T±2sT/log N0+0.2507).
Replacement of Missing Values
If not more than one specimen from a group and not more than two specimens from all of the groups giving the results f1through fkare missing,replace each missing value by adding 0to the number showing no growth,if the number showing no growth in the remaining nine specimens of that group is 4or less,and adding 1if the number showing no growth in the remaining nine specimens of that group is 5or more.
Survival Time and Kill Time
Take two groups,each consisting of 10specimens of the relevant biological indicator,in their original,individual containers.Place the specimens of a group in suitable specimen holders that permit each specimen to be exposed to the sterilizing conditions at a specific location in the sterilizing chamber.Check the chamber for operating parameters by preheating it to the selected temperature ±2 in the cases of Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,or ±0.5
in the cases of Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,or ±0.5 in the cases of Biological Indicator for Steam Sterilization,Paper Carrier,and Biological Indicator for Steam Sterilization,Self-Contained.
in the cases of Biological Indicator for Steam Sterilization,Paper Carrier,and Biological Indicator for Steam Sterilization,Self-Contained.
For Biological Indicator for Dry-Heat Sterilization,Paper Carrier,preheat the unit to temperature,and equilibrate the heat chamber.Open the access door or port,and place the holder(s)in the chamber,close the access door or port,and continue to operate the apparatus.Commence timing the heat exposure when the chamber temperature returns to the lower limit of the selected temperature.Expose the specimens for the required survival time,enter the chamber,and remove the holder(s)containing the 10specimens.Repeat the above procedure immediately,or preheat if a substantial interval has elapsed,so as to subject the second holder(s)containing 10specimens similarly to the first conditions,but for the required kill time.
For Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,preheat the chamber to equilibrium at the selected temperature ±2 ,and initiate and monitor the operating steps 1through 6as described for D Value Determination,appropriate for the combination of gas concentration,temperature,and relative humidity,using a gassing time in step 4appropriate to the survival time.Repeat the above procedure with two or more groups each consisting of 10specimens,but using a gassing time in step 4appropriate to the kill time.
,and initiate and monitor the operating steps 1through 6as described for D Value Determination,appropriate for the combination of gas concentration,temperature,and relative humidity,using a gassing time in step 4appropriate to the survival time.Repeat the above procedure with two or more groups each consisting of 10specimens,but using a gassing time in step 4appropriate to the kill time.
For Biological Indicator for Steam Sterilization,Paper Carrier,and Biological Indicator for Steam Sterilization,Self-Contained,exhaust the steam chamber,and open the door.Within 15seconds of opening the door place the loaded holder(s)into the chamber,and operate the apparatus to heat the chamber contents as quickly as possible.Expose the specimens for the required survival time,counting the exposure from the time when the temperature record shows that the chamber has reached the required temperature.Exhaust the chamber as quickly as possible at the end of the exposure period.When the chamber can be safely entered,remove the holder(s)containing the specimens.Repeat the above procedure immediately,or preheat if a substantial interval has elapsed,so as to subject the holder(s)containing 10specimens similarly to the first exposure.Repeat the above procedure with two more groups each consisting of 10specimens,but expose the specimens for the required kill time.In each case for the Biological Indicator for Dry-Heat Sterilization,Paper Carrier;Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier;or Biological Indicator for Steam Sterilization,Paper Carrier,whichever is appropriate,after completion of the sterilizing procedure,and within a noted time not more than 4hours,aseptically remove and add each carrier to 10to 30mLof Soybean–Casein Digest Medium(see Mediaunder Sterility Tests á71ñ)to submerge the biological indicator completely in a suitable tube.Incubate each tube at a temperature of 55 to 60
to 60 in the case of Biological Indicator for Steam Sterilization,Paper Carrier,or of 30
in the case of Biological Indicator for Steam Sterilization,Paper Carrier,or of 30 to 35
to 35 in the cases of Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,or at the optimal temperature specified by the manufacturer.For Biological Indicator for Steam Sterilization,Self-Contained,the paper strip is immersed in self-contained medium according to manufacturers'instructions within a noted time not more than 4hours and incubated at 55
in the cases of Biological Indicator for Dry-Heat Sterilization,Paper Carrier,and Biological Indicator for Ethylene Oxide Sterilization,Paper Carrier,or at the optimal temperature specified by the manufacturer.For Biological Indicator for Steam Sterilization,Self-Contained,the paper strip is immersed in self-contained medium according to manufacturers'instructions within a noted time not more than 4hours and incubated at 55 to 60
to 60 .Observe each inoculated medium-containing tube at 24and 48hours,and every 1or 2days thereafter for a total of 7days after inoculation.(Where growth is observed at any particular observation time,further incubation of the specimen(s)concerned may be omitted.)Note the specimens showing no evidence of growth at any time.
.Observe each inoculated medium-containing tube at 24and 48hours,and every 1or 2days thereafter for a total of 7days after inoculation.(Where growth is observed at any particular observation time,further incubation of the specimen(s)concerned may be omitted.)Note the specimens showing no evidence of growth at any time.
1
Safety includes design to prevent electric shock or gas exposition and burns,where operators can wear protective clothing and gloves against burns from touching hot surfaces.
2
Descriptions of different types of dry-heat sterilizing equipment and detailed guidelines for determining,monitoring,and controlling the operating parameters have been published by the Health Industry Manufacturers Association in Report No.78-1.7,Operator Training for Dry Heat Sterilizing Equipment,and by the Parenteral Drug Association,Inc.,in Technical Report No.3,Validation of Dry Heat Processes Used for Sterilization and Depyrogenation.
3
Standard for BIER/EO Gas Vessels,1July 1992,Association for the Advancement of Medical Instrumentation (AAMI),3330Washington Boulevard,Suite 440,Arlington,VA22201-4598.
4
Standard for BIER/Steam Vessels,1July 1992,AAMI,3330Washington Boulevard,Suite 400,Arlington,VA22201-4598.