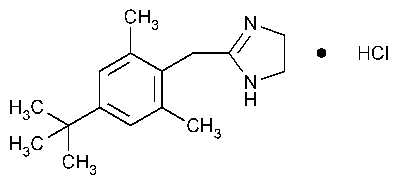
Xylometazoline Hydrochloride
1H-Imidazole,2-[[4-(1,1-dimethylethyl)-2,6-dimethylphenyl]methyl]-4,5-dihydro-,monohydrochloride.
2-(4-tert-Butyl-2,6-dimethylbenzyl)-2-imidazoline monohydrochloride [1218-35-5].
»Xylometazoline Hydrochloride contains not less than 99.0percent and not more than 101.0percent of C16H24N2·HCl,calculated on the dried basis.
Packaging and storage—
Preserve in tight,light-resistant containers.
Identification—
A:
Infrared Absorption á197Mñ.
B:
The RFvalue of the principal spot in the chromatogram of the Identification preparationcorresponds to that of Standard preparation Aas obtained in the test for Chromatographic purity.
pHá791ñ:
between 5.0and 6.6,in a solution (1in 20).
Loss on drying á731ñ—
Dry it at 105 for 4hours:it loses not more than 0.5%of its weight.
for 4hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Chromatographic purity—
Standard solutions—
Dissolve USP Xylometazoline Hydrochloride RSin methanol,and mix to obtain Standard preparation Ahaving a known concentration of 100µg per mL.Dilute quantitatively with methanol to obtain Standard solutions,designated below by letter,having the following compositions:
| Standardsolution | Dilution | Concentration (µg RSper mL) |
Percentage (%, for comparison with test specimen) |
| A | (undiluted) | 100 | 0.5 |
| B | (4in 5) | 80 | 0.4 |
| C | (3in 5) | 60 | 0.3 |
| D | (2in 5) | 40 | 0.2 |
| E | (1in 5) | 20 | 0.1 |
Test solution—
Dissolve an accurately weighed quantity of Xylometazoline Hydrochloride in methanol to obtain a solution containing 20mg per mL.
Identification solution—
Dilute a portion of the Test solutionquantitatively with methanol to obtain a solution containing 100µg per mL.
Detection reagent—
Prepare (1)a solution of 0.5g of potassium iodide in 50mLof water,and (2)a solution of 1.5g of soluble starch in 50mLof boiling water.Just prior to use,mix 10mLof each solution with 3mLof alcohol.
Procedure—
Apply separately 5µLof the Test solution,5µLof the Identification solution,and 5µLof each Standard solutionto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel.Position the plate in a chromatographic chamber,and develop the chromatograms in a solvent system consisting of a mixture of methanol and ammonium hydroxide (20:1)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the developing chamber,mark the solvent front,and allow the plate to dry under a current of warm air for at least 30minutes.Expose the plate to chlorine gas for not more than 5minutes,and air-dry until the chlorine has dissipated (about 15minutes).Spray the plate with Detection reagent,and immediately compare the intensities of any secondary spots observed in the chromatogram of the Test solutionwith those of the principal spots in the chromatograms of the Standard solutions:the sum of the intensities of all secondary spots obtained from the Test solutioncorresponds to not more than 1.0%.
Assay—
Dissolve about 500mg of Xylometazoline Hydrochloride,accurately weighed,in 70mLof glacial acetic acid,add 10mLof mercuric acetate TS,and titrate with 0.1Nperchloric acid VS,determining the endpoint potentiometrically (see Titrimetry á541ñ),using a calomel-glass electrode system.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 28.08mg of C16H24N2·HCl.
Auxiliary Information—
Staff Liaison:Kahkashan Zaidi,Ph.D.,Senior Scientific Associate
Expert Committee:(AER)Aerosols
USP28–NF23Page 2041
Phone Number:1-301-816-8269