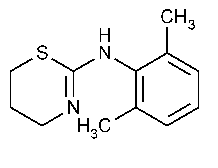
Xylazine
»Xylazine contains not less than 98.0percent and not more than 102.0percent of C12H16N2S.
Packaging and storage—
Preserve in tight containers.Store at 25 ,excursions permitted between 15
,excursions permitted between 15 and 30
and 30 .
.
Labeling—
Where it is intended for veterinary use only,the label so states.
Identification—
C:Thin-Layer Chromatographic Identification Test á201ñ—
Test solution:
2mg per mL,in chloroform.
Developing solvent system:
acetone,chloroform,and methanol (2:1:1).
Procedure—
Prior to the applications of theTest solution and the Standard solution,dry the plate at 105 for not less than 30minutes,and allow it to cool in a desiccator.Allow the applications to dry with the aid of a current of warm air,and develop.Examine under short-wavelength UVlight:the size,intensity,and RFvalue of the principal spot obtained from theTest solution correspond to those of the principal spot obtained from theStandard solution.
for not less than 30minutes,and allow it to cool in a desiccator.Allow the applications to dry with the aid of a current of warm air,and develop.Examine under short-wavelength UVlight:the size,intensity,and RFvalue of the principal spot obtained from theTest solution correspond to those of the principal spot obtained from theStandard solution.
Loss on drying á731ñ—
Dry it in vacuum at 60 for 4hours:it loses not more than 0.5%of its weight.
for 4hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Heavy metals,Method IIá231ñ:
20µg per g.
Limit of 3-amino-1-propanol—
Prepare a test solution of Xylazine in methanol containing 100mg per mL,using sonication to achieve dissolution.Prepare a Standard solution of 3-amino-1-propanol in methanol containing 0.5mg per mL.Separately apply 5µLof the test solution and the Standard solution to a thin-layer chromatographic plate (seeChromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel.Allow the applications to dry,and develop the chromatograms in a saturated chromatographic chamber,containing a solvent system consisting of a mixture of alcohol and ammonium hydroxide (80:20)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the chromatographic chamber,mark the solvent front,and air-dry the plate.Spray the plate with an alcoholic solution of ninhydrin (1in 500),and immediately heat the plate in an oven at 105 .When the spots are visible,remove the plate from the oven,and allow to cool.Examine the chromatograms,and compare the intensities of the spots corresponding to 3-amino-1-propanol:the intensity of the spot for 3-amino-1-propanol obtained from the test solution is not greater than that of the spot for 3-amino-1-propanol obtained from the Standard solution (0.5%).
.When the spots are visible,remove the plate from the oven,and allow to cool.Examine the chromatograms,and compare the intensities of the spots corresponding to 3-amino-1-propanol:the intensity of the spot for 3-amino-1-propanol obtained from the test solution is not greater than that of the spot for 3-amino-1-propanol obtained from the Standard solution (0.5%).
Limit of acetone and isopropyl alcohol—
Diluent—
Dilute 15mLof glacial acetic acid with water to 1000mL,and mix.
Standard solution—
Transfer 10.0µLeach of acetone and isopropyl alcohol to a 500-mLvolumetric flask,dilute withDiluentto volume,and mix.This solution contains 15.8µg of acetone per mLand 15.7µg of isopropyl alcohol per mL.
Test solution—
Transfer about 100mg of Xylazine,accurately weighed,to a 10-mLvolumetric flask,dissolve in and dilute withDiluentto volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The gas chromatograph is equipped with a flame-ionization detector and a 2-mm ×1.8-m column packed with 0.1%phase G25on 80-to 100-mesh support S7.Helium is used as the carrier gas with a flow rate of about 30mLper minute.The injection port and detector temperatures are maintained at about 240 and 275
and 275 ,respectively.The system is programmed according to the following steps.The column temperature is maintained at 30
,respectively.The system is programmed according to the following steps.The column temperature is maintained at 30 for 6minutes after each injection,then increased to 100
for 6minutes after each injection,then increased to 100 at a rate of 10
at a rate of 10 per minute,then increased further to 220
per minute,then increased further to 220 at a rate of 15
at a rate of 15 per minute,and maintained for 10minutes.Chromatograph theStandard solution,and record the peak responses as directed forProcedure:the relative retention times are about 0.75for acetone and 1.0for isopropyl alcohol;the resolution,R,between acetone and isopropyl alcohol is not less than 2.0;the tailing factor determined from each analyte peak is not more than 2.0;and the relative standard deviation for replicate injections is not more than 2.0%.
per minute,and maintained for 10minutes.Chromatograph theStandard solution,and record the peak responses as directed forProcedure:the relative retention times are about 0.75for acetone and 1.0for isopropyl alcohol;the resolution,R,between acetone and isopropyl alcohol is not less than 2.0;the tailing factor determined from each analyte peak is not more than 2.0;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 2µL)of theStandard solutionand theTest solutioninto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the percentages of acetone and isopropyl alcohol in the portion of Xylazine taken by the formula:
(C/W)(rU/rS),
in whichCis the concentration,in µg per mL,of acetone or isopropyl alcohol in each mLof theStandard solution;Wis the weight,in mg,of Xylazine taken to prepare theTest solution;andrUandrSare the responses for the relevant analyte peak obtained from theTest solution and theStandard solution,respectively:not more than 0.02%of acetone and not more than 0.2%of isopropyl alcohol are found.
Chromatographic purity—
Solution A,Solution B,Mobile phase,and Diluent—
Proceed as directed in theAssay.
Standard solution—
Quantitatively dilute an accurately measured volume of theStandard preparationprepared in theAssaywithDiluentto obtain a solution having a concentration of 0.008mg ofUSP Xylazine RSper mL.
Test solution—
Transfer about 100mg of Xylazine,accurately weighed,to a 10-mLvolumetric flask,add 5.0mLofSolution B,and swirl to dissolve.Add about 4mLofSolution A,and swirl.Dilute withSolution Ato volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 205-nm detector and a 4.6-mm ×25-cm column that contains packing L7and a guard column.The flow rate is about 1mLper minute.Equilibrate the column with a mobile phase consisting of 75%Solution Aand 25%Solution B.Maintain this composition for 8minutes following each injection,after which the proportion ofSolution Bis increased linearly from 25%to 70%over a period of 27minutes,and maintained at that composition for 5minutes;then rapidly increase the proportion ofSolution Ato 75%before the next injection.Chromatograph theStandard solution,and record the peak responses as directed forProcedure:the tailing factor is not more than 1.5;and the relative standard deviation for replicate injections is not more than 5.0%.
Procedure—
Separately inject equal volumes (about 10µL)of theStandard solutionand theTest solutioninto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the percentage of each impurity in the Xylazine taken by the formula:
1000(C/W)(riF/rS),
in whichCis the concentration,in mg per mL,of USP Xylazine RSin theStandard solution;Wis the weight,in mg,of Xylazine taken to prepare theTest solution;riis the response of any individual impurity peak in the chromatogram of theTest solution that is not present in the chromatogram of theDiluent;Fis the response factor of 0.72for the 2,6-dimethylaniline peak at a response time of about 0.8relative to the retention time of xylazine,of 0.36for an impurity at a relative retention time of about 1.3,0.37for 2,6-dimethylphenyl isothiocyanate at a relative retention time of about 2,and 1.0for any other impurity;andrSis the response of the xylazine peak in the chromatogram of theStandard solution:not more than 0.5%of any individual impurity is found;and the sum of all impurities found is not more than 1%.
Assay—
Solution A—
Dissolve 3.03g of sodium 1-heptanesulfonate in 800mLof water,adjust with 2Nsulfuric acid to a pHof 3.0,dilute with water to 1000mL,and mix.Pass through a filter having a 0.5-µm or finer porosity.
Solution B—
Use acetonitrile.
Mobile phase—
Use variable mixtures ofSolution AandSolution Bas directed forChromatographic system.
Diluent—
Prepare a mixture ofSolution AandSolution B(50:50).
Standard preparation—
Prepare a solution of USP Xylazine RSinDiluenthaving a known concentration of about 0.4mg per mL.
Assay preparation—
Transfer about 10mg of Xylazine,accurately weighed,to a 25-mLvolumetric flask,dilute withDiluentto volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 226-nm detector and a 3.9-mm ×30-cm column that contains packing L1.The flow rate is about 1mLper minute.Equilibrate the column with a mobile phase consisting of 70%Solution Aand 30%Solution B.Maintain this composition for 5minutes following each injection,after which the proportion ofSolution Bis increased linearly from 30%to 40%over a period of 5minutes,and maintained at that composition for 5minutes;then rapidly increase the proportion ofSolution Ato 70%before the next injection.Chromatograph theStandard preparation,and record the peak responses as directed forProcedure:the tailing factor is not more than 2.0;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10µL)of theStandard preparationand theAssay preparationinto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the quantity,in mg,of C12H16N2Sin the portion of Xylazine taken by the formula:
25C(rU/rS),
in whichCis the concentration,in mg per mL,of USP Xylazine RSin theStandard preparation;andrUandrSare the xylazine peak responses obtained from theAssay preparation and theStandard preparation,respectively.
Auxiliary Information—
Staff Liaison:Ian DeVeau,Ph.D.,Senior Scientist
Expert Committee:(VET)Veterinary Drugs
USP28–NF23Page 2038
Pharmacopeial Forum:Volume No.29(6)Page 2004
Phone Number:1-301-816-8178