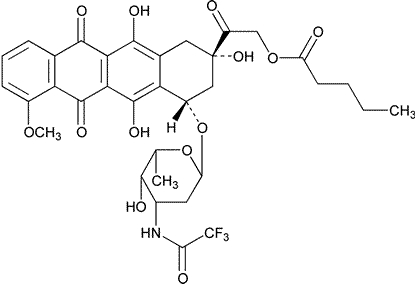
Valrubicin
C34H36F3NO13
723.65
(2S-cis)-2-[1,2,3,4,6,11-Hexahydro-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-4-[[2,3,6-trideoxy-3-[(trifluoroacetyl)amino]-a-L-lyxo-hexopyranosyl]oxy]-2-naphthacenyl]-2-oxoethyl pentanoate.
(8S,10S)-8-Glycoloyl-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-10-[[2,3,6-trideoxy-3-(2,2,2-trifluoroacetamido)-a-L-lyxo-hexopyranosyl]oxy]-5,12-naphthacenedione 82-valerate [56124-62-0].
(2S-cis)-2-[1,2,3,4,6,11-Hexahydro-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-4-[[2,3,6-trideoxy-3-[(trifluoroacetyl)amino]-a-L-lyxo-hexopyranosyl]oxy]-2-naphthacenyl]-2-oxoethyl pentanoate.
(8S,10S)-8-Glycoloyl-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-10-[[2,3,6-trideoxy-3-(2,2,2-trifluoroacetamido)-a-L-lyxo-hexopyranosyl]oxy]-5,12-naphthacenedione 82-valerate [56124-62-0].
»Valrubicin contains not less than 95.0percent and not more than 103.0percent of C34H36F3NO13,calculated on the dried basis.
Caution—Great care should be taken to prevent inhaling particles of Valrubicin and exposing the skin to it.
Packaging and storage—
Preserve in tight,light-resistant containers at controlled room temperature.
Identification—
A:
Infrared Absorption á197Mñ.
B:
Ultraviolet Absorption á197Uñ—
Solution:
10mg per mL.
Medium:
methanol.
Absorptivities,calculated on the dried basis,are 555±20at 233nm and 382.5±17.5at 252nm.
C:
The retention time of the major peak in the chromatogram of the Assay preparationcorresponds to that in the chromatogram of the Standard preparation,as obtained in the Assay.
Loss on drying á731ñ—
Dry it in vacuum over phosphorus pentoxide at 80 for 4hours:it loses not more than 3.0%of its weight.
for 4hours:it loses not more than 3.0%of its weight.
Residue on ignition á281ñ:
not more than 0.2%.
Limit of residual solvents—
Internal standard solution—
Prepare a solution of n-propyl alcohol in methyl sulfoxide having a concentration of about 0.05µLper mL.
Standard solution—
Prepare a solution in Internal standard solutionhaving a concentration of 2.5µg of chloroform,5.0µg of dehydrated alcohol,5.0µg of acetone,5.0µg of butyl alcohol,5.0µg of dioxane,10.0µg of methylene chloride,15.0µg of diisopropyl ether,20.5µg of acetonitrile,50µg of pentane,and 100µg of methanol in each mL,and sonicate.
Test solution—
Dissolve about 200mg of Valrubicin,accurately weighed,in 4.0mLof Internal standard solution,and sonicate.
Chromatographic system (see Chromatography á621ñ)—
The gas chromatograph is equipped with a flame-ionization detector and a 0.32-mm ×30-m fused-silica capillary column coated with a 5-µm film of G2stationary phase.The carrier gas is helium,flowing at a rate of 30mLper minute.The column temperature is maintained at 220 .The injection port temperature and the detector block temperature are maintained at 250
.The injection port temperature and the detector block temperature are maintained at 250 .Chromatograph the Standard solution,and record the responses as directed for Procedure:the component solvent peaks are resolved;the relative retention times are about 0.48for methanol,0.66for dehydrated alcohol,0.71for acetonitrile,0.76for acetone,0.86for pentane,0.92for methylene chloride,1.0for n-propyl alcohol,1.19for diisopropyl ether,1.22for chloroform,1.35for butyl alcohol,and 1.52for dioxane;and the relative standard deviation of the ratios of the peak area of each solvent to the peak area of n-propyl alcohol is not more than 10%.
.Chromatograph the Standard solution,and record the responses as directed for Procedure:the component solvent peaks are resolved;the relative retention times are about 0.48for methanol,0.66for dehydrated alcohol,0.71for acetonitrile,0.76for acetone,0.86for pentane,0.92for methylene chloride,1.0for n-propyl alcohol,1.19for diisopropyl ether,1.22for chloroform,1.35for butyl alcohol,and 1.52for dioxane;and the relative standard deviation of the ratios of the peak area of each solvent to the peak area of n-propyl alcohol is not more than 10%.
Procedure—
Separately inject equal volumes (about 1µL)of the Standard solutionand the Test solutioninto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the concentration,in µg per g,of each residual solvent in the portion of Valrubicin taken by the formula:
4000(C/W)(Ri/RS),
in which Cis the concentration,in µg per mL,of the respective individual solvent in the Standard solution;Wis the quantity,in mg,of Valrubicin taken to prepare the Test solution;and Riand RSare the peak area ratios of the respective individual solvent to n-propyl alcohol obtained from the Test solutionand the Standard solution,respectively:not more than 50µg per g of chloroform,100µg per g of dehydrated alcohol,100µg per g of acetone,100µg per g of butyl alcohol,100µg per g of dioxane,300µg per g of methylene chloride,410µg per g of acetonitrile,500µg per g of diisopropyl ether,1000µg per g of pentane,and 2000µg per g of methanol are found.
Related compounds—
Mobile phase—
Prepare as directed in the Assay.
Resolution solution—
Prepare a solution of USP Valrubicin Related Compound A RSand USP Valrubicin RSin acetonitrile having known concentrations of about 0.25mg per mLand 1mg per mL,respectively.
Test solution—
Use the Assay preparation.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 254-nm detector,a guard column,and a 5-mm ×10-cm analytical column that contains a 4-µm packing L1.The flow rate is about 3.5mLper minute.Chromatograph the Resolution solution,and record the responses as directed for Procedure:the relative retention times are about 0.8for valrubicin related compound Aand 1.0for valrubicin;and the resolution,R,between valrubicin related compound Aand valrubicin is not less than 2.
Procedure—
Inject a volume (about 10µL)of the Test solutioninto the chromatograph,record the chromatogram,and measure the areas for the major peaks.Calculate the percentage of each impurity in the portion of Valrubicin taken by the formula:
100(ri/rs),
in which riis the peak area for each impurity;and rsis the sum of the areas of all the peaks.Do not consider any peaks due to solvent or excipients.Not more than 0.3%of any individual impurity with a relative retention time of 0.06,0.17,0.27,or 0.52is found;not more than 0.6%of any impurity with a relative retention time of about 0.14is found;not more than 0.2%of any other individual impurity is found;not more than 1.0%of total other impurities that are not specified by relative retention time is found;and not more than 2.5%of total impurities that are not less than 0.1%is found.
Assay—
Mobile phase—
Prepare a filtered and degassed mixture of 0.015Mphosphoric acid and acetonitrile (57:43).Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Valrubicin RSin acetonitrile,and dilute quantitatively with acetonitrile to obtain a solution having a known concentration of about 1mg per mL.
Assay preparation—
Transfer about 25mg of Valrubicin,accurately weighed,to a 25-mLvolumetric flask,dissolve in and dilute with acetonitrile to volume,and mix.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 254-nm detector,a guard column,and a 5-mm ×10-cm analytical column that contains a 4-µm packing L1.The flow rate is about 3.5mLper minute.Chromatograph the Standard preparation,and record the responses as directed for Procedure:the relative standard deviation for replicate injections is not more than 2%.
Procedure—
Separately inject equal volumes (about 10µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the quantity,in mg,of C34H36F3NO13in the portion of Valrubicin taken by the formula:
0.25CP(rU/rS),
in which Cis the concentration,in mg per mL,of USP Valrubicin RSin the Standard preparation;Pis the specified percentage of valrubicin in USP Valrubicin RS;and rUand rSare the valrubicin peak areas obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 2011
Pharmacopeial Forum:Volume No.30(3)Page 946
Phone Number:1-301-816-8335