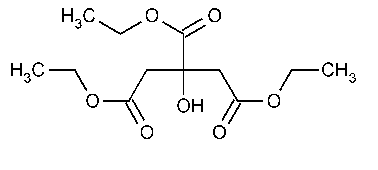
Triethyl Citrate
»Triethyl Citrate contains not less than 99.0percent and not more than 100.5percent of C12H20O7,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
Identification—
B:
The retention time of the major peak in the chromatogram of theAssay preparation corresponds to that in the chromatogram of a similar preparation of USP Triethyl Citrate RS.
Specific gravity á841ñ:
between 1.135and 1.139.
Refractive index á831ñ:
between 1.439and 1.441.
Acidity—
Dissolve 32.0g in 30mLof alcohol,previously neutralized to phenolphthalein,add bromothymol blue TS,and titrate with 0.10Nsodium hydroxide.Not more than 1.0mLis required.
Water,Method Iá921ñ:
not more than 0.25%.
Heavy metals,Method IIá231ñ:
0.001%.
Assay—
System suitability solution—
Prepare a solution in toluene containing about 30mg each of USP Triethyl Citrate RSand USP Acetyltriethyl Citrate RSper mL.
Assay preparation—
Transfer about 300mg of Triethyl Citrate,accurately weighed,to a 10-mLvolumetric flask,dissolve in and dilute with toluene to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The gas chromatograph is equipped with an on-column,temperature-programmable injector,a flame-ionization detector maintained at about 275 ,and a 0.32-mm ×30-m column bonded with a 0.5-µm layer of phase G42.The column temperature is programmed to be maintained at about 80
,and a 0.32-mm ×30-m column bonded with a 0.5-µm layer of phase G42.The column temperature is programmed to be maintained at about 80 for 0.5minute,then to increase to about 220
for 0.5minute,then to increase to about 220 at a rate of 20
at a rate of 20 per minute,and to hold at about 220
per minute,and to hold at about 220 for 10minutes.The injection port temperature is programmed to be maintained at about 85
for 10minutes.The injection port temperature is programmed to be maintained at about 85 for 0.5minute,then to increase to about 225
for 0.5minute,then to increase to about 225 at a rate of 20
at a rate of 20 per minute,and to hold at about 225
per minute,and to hold at about 225 for 10minutes.Helium is used as the carrier gas at a flow rate of about 2.3mLper minute.Chromatograph theSystem suitability solution,and record the peak responses as directed forProcedure:the relative retention times are about 0.9for triethyl citrate and 1.0for acetyltriethyl citrate;the resolution,R,between triethyl citrate and acetyltriethyl citrate is not less than 1.5;and the relative standard deviation for replicate injections is not more than 2.0%determined from both the triethyl citrate and acetyltriethyl citrate peaks,based on area percent calculation.
for 10minutes.Helium is used as the carrier gas at a flow rate of about 2.3mLper minute.Chromatograph theSystem suitability solution,and record the peak responses as directed forProcedure:the relative retention times are about 0.9for triethyl citrate and 1.0for acetyltriethyl citrate;the resolution,R,between triethyl citrate and acetyltriethyl citrate is not less than 1.5;and the relative standard deviation for replicate injections is not more than 2.0%determined from both the triethyl citrate and acetyltriethyl citrate peaks,based on area percent calculation.
Procedure—
[NOTE—Use peak areas where peak responses are indicated.]Inject 1µLof the Assay preparationinto the chromatograph,record the chromatogram,and measure all of the peak areas,excluding the solvent peak.Calculate the percentage of C12H20O7in the portion of Triethyl Citrate taken by the formula:
100(A/B),
in whichAis the triethyl citrate peak response;andBis the sum of the responses of all the peaks.
Auxiliary Information—
Staff Liaison:Ravi Ravichandran,Ph.D.,Senior Scientist
Expert Committee:(EMC)Excipients:Monograph Content
USP28–NF23Page 3101
Pharmacopeial Forum:Volume No.27(5)Page 3068
Phone Number:1-301-816-8330