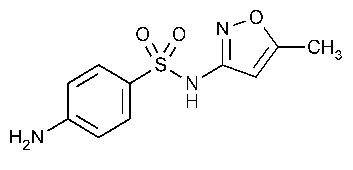
Sulfamethoxazole
Benzenesulfonamide,4-amino-N-(5-methyl-3-isoxazolyl)-.
N1-(5-Methyl-3-isoxazolyl)sulfanilamide [723-46-6].
»Sulfamethoxazole contains not less than 99.0percent and not more than 101.0percent of C10H11N3O3S,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed,light-resistant containers.Store at 25 ,excursions permitted between 15
,excursions permitted between 15 and 30
and 30 .
.
Identification—
A:Infrared Absorption á197Kñ.
B:Ultraviolet Absorption á197Uñ—
Solution:
10µg per mL.
Medium:
sodium hydroxide solution (1in 250).
Absorptivities at 257nm,calculated on the dried basis,do not differ by more than 2.0%.
C:
Dissolve about 100mg in 2mLof hydrochloric acid,and add 3mLof sodium nitrite solution (1in 100)and 1mLof sodium hydroxide solution (1in 10)containing 10mg of 2-naphthol:a red-orange precipitate is formed.
Loss on drying á731ñ—
Dry it at 105 for 4hours:it loses not more than 0.5%of its weight.
for 4hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Selenium á291ñ:
0.003%,a 200-mg test specimen being used.
Sulfanilamide and sulfanilic acid—
Standard solution—
Dissolve 100mg of USP Sulfamethoxazole RSin 0.10mLof ammonium hydroxide,dilute with methanol to 10.0mL,and mix.
Reference solution—
Dissolve 20mg of USP Sulfanilamide RSand 20mg of sulfanilic acid in 10mLof ammonium hydroxide,and dilute with methanol to 100mL.Transfer 2.0mLof the solution to a 50-mLvolumetric flask,add 10mLof ammonium hydroxide,dilute with methanol to volume,and mix.
Test solution—
Dissolve 100mg in 0.10mLof ammonium hydroxide,dilute with methanol to 10.0mL,and mix.
Procedure—
Apply 10µLof the Standard solution,25µLof the Reference solution,and 10µLof the Test solutionto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel.Allow the spots to dry,and develop the chromatogram in a solvent system consisting of a mixture of alcohol,n-heptane,chloroform,and glacial acetic acid (25:25:25:7)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the chamber,and allow it to air-dry.Spray the plate with a solution prepared by dissolving 0.10g of p-dimethylaminobenzaldehyde in 1mLof hydrochloric acid and diluting with alcohol to 100mL.Sulfamethoxazole produces a spot at about RF0.7,sulfanilamide at about RF0.5,and sulfanilic acid at about RF0.1.Any spots produced by sulfanilamide or sulfanilic acid from the Test solutiondo not exceed in size or intensity similar spots,occurring at the respective RFvalues,produced by sulfanilamide or sulfanilic acid from the Reference preparation(0.2%).
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Dissolve about 500mg of Sulfamethoxazole,accurately weighed,in a mixture of 20mLof glacial acetic acid and 40mLof water,and add 15mLof hydrochloric acid.Cool to 15 ,and immediately titrate with 0.1Msodium nitrite VS,determining the endpoint potentiometrically using a calomel-platinum electrode system.Each mLof 0.1Msodium nitrite is equivalent to 25.33mg of C10H11N3O3S.
,and immediately titrate with 0.1Msodium nitrite VS,determining the endpoint potentiometrically using a calomel-platinum electrode system.Each mLof 0.1Msodium nitrite is equivalent to 25.33mg of C10H11N3O3S.
Auxiliary Information—
Staff Liaison:Behnam Davani,Ph.D.,MBA,Senior Scientist
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 1828
Pharmacopeial Forum:Volume No.29(6)Page 1989
Phone Number:1-301-816-8394