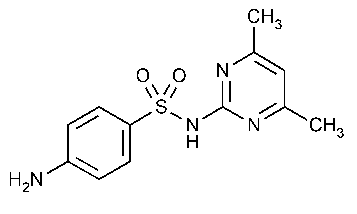
Sulfamethazine
Benzenesulfonamide,4-amino-N-(4,6-dimethyl-2-pyrimidinyl)-.
N1-(4,6-Dimethyl-2-pyrimidinyl)sulfanilamide [57-68-1].
»Sulfamethazine contains not less than 99.0percent and not more than 100.5percent of C12H14N4O2S,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed,light-resistant containers.
Clarity and color of solution—
Dissolve 1.0g in a mixture of 20mLof water and 5mLof 1Nsodium hydroxide:the solution is clear and not more deeply colored than pale yellow.
Identification—
A:
Infrared Absorption á197Kñ.
B:
To 0.10g add 10mLof water and just sufficient sodium hydroxide solution (1in 250)to give a faint pink spot on phenolphthalein paper.Add 5drops of cupric sulfate TS:a yellow-green precipitate is formed,and it becomes brown on standing.
Acidity—
Digest 3.0g with 150mLof carbon dioxide-free water at 70 for about 5minutes,stirring occasionally to maintain suspension of the test specimen.Cool the mixture rapidly in an ice bath to 20±0.5
for about 5minutes,stirring occasionally to maintain suspension of the test specimen.Cool the mixture rapidly in an ice bath to 20±0.5 ,with mechanical stirring.Filter immediately,with vacuum,omitting washing of the solid but drying it thoroughly by suction.Add 2drops of thymolphthalein TSto 25.0mLof the filtrate,and titrate with 0.1Nsodium hydroxide VS.To a second 25.0-mLportion of the filtrate add 10mLof hydrochloric acid.Cool the mixture to 15
,with mechanical stirring.Filter immediately,with vacuum,omitting washing of the solid but drying it thoroughly by suction.Add 2drops of thymolphthalein TSto 25.0mLof the filtrate,and titrate with 0.1Nsodium hydroxide VS.To a second 25.0-mLportion of the filtrate add 10mLof hydrochloric acid.Cool the mixture to 15 ,and titrate with 0.1Msodium nitrite VSas directed under Nitrite Titration á451ñ:the volume of 0.1Nsodium hydroxide consumed does not exceed the volume of 0.1Msodium nitrite consumed by more than 0.5mL.
,and titrate with 0.1Msodium nitrite VSas directed under Nitrite Titration á451ñ:the volume of 0.1Nsodium hydroxide consumed does not exceed the volume of 0.1Msodium nitrite consumed by more than 0.5mL.
Loss on drying á731ñ—
Dry about 1g,accurately weighed,at 105 for 2hours:it loses not more than 0.5%of its weight.
for 2hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Selenium á291ñ:
0.003%,a 200-mg test specimen being used.
Heavy metals,Method IIá231ñ:
0.002%.
Ordinary impurities á466ñ—
Test solution:
acetone.
Standard solution:
acetone.
Eluant:
a mixture of ethyl acetate,methanol,and ammonium hydroxide (17:6:5).
Visualization:
11.
Assay—
Proceed with Sulfamethazine as directed under Nitrite Titration á451ñ.Each mLof 0.1Msodium nitrite is equivalent to 27.83mg of C12H14N4O2S.
Auxiliary Information—
Staff Liaison:Behnam Davani,Ph.D.,MBA,Senior Scientist
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 1826
Phone Number:1-301-816-8394