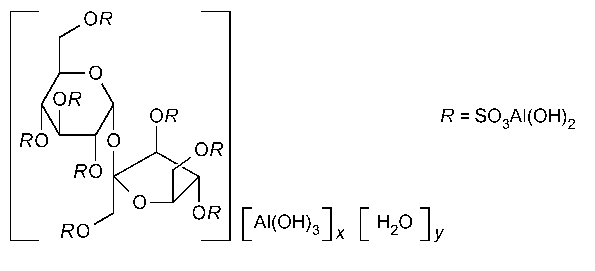
Sucralfate
»Sucralfate is the hydrous basic aluminum salt of sucrose octasulfate.It contains the equivalent of not less than 30.0percent and not more than 38.0percent of sucrose octasulfate (C12H14O35S8).
Packaging and storage—
Preserve in tight containers.
USP Reference standards á11ñ—
USP Potassium Sucrose Octasulfate RS.
Clarity and color of solution—
Dissolve 1.0g in 10mLof 2Nsulfuric acid:the solution is clear and practically colorless.
Identification—
A:
The retention time of the sucrose octasulfate peak in the chromatogram of theAssay preparation corresponds to that in the chromatogram of theStandard preparation,as obtained in theAssay.
B:
Add 0.1Nhydrochloric acid to a few g of Sucralfate,boil,and neutralize with 0.1Nsodium hydroxide.Add alkaline cupric tartrate TS.Boil a small amount of this solution:a red precipitate of cuprous oxide is produced.
C:
Asolution in 3Nhydrochloric acid meets the requirements of the tests for Aluminum á191ñ.
Acid-neutralizing capacity—
Transfer about 250mg,accurately weighed,to a 250-mLscrew-capped bottle,add 100.0mLof 0.1Nhydrochloric acid,previously heated to 37 ,cap the bottle,place it in a 37
,cap the bottle,place it in a 37 water bath,and stir the contents continuously for 1hour.Cool to room temperature,and transfer 20.0mLto a 100-mLbeaker.Add 30mLof water,and titrate with with 0.1Nsodium hydroxide VSto a pHof 3.5.Perform a blank titration on a mixture of water and 0.1Nhydrochloric acid (30:20.0).Calculate the mEq of acid consumed per g of Sucralfate taken by the formula:
water bath,and stir the contents continuously for 1hour.Cool to room temperature,and transfer 20.0mLto a 100-mLbeaker.Add 30mLof water,and titrate with with 0.1Nsodium hydroxide VSto a pHof 3.5.Perform a blank titration on a mixture of water and 0.1Nhydrochloric acid (30:20.0).Calculate the mEq of acid consumed per g of Sucralfate taken by the formula:
5N(VB–VT)/W,
whereNis the exact normality of the sodium hydroxide VS;VBandVTare the volumes,in mL,of sodium hydroxide VSconsumed by the blank and the test solution,respectively;andWis the weight,in g,of Sucralfate taken:not less than 12mEq of acid is consumed.
Chloride á221ñ—
Transfer 500mg to a 100-mLvolumetric flask,add 30mLof 2Nnitric acid,dilute with water to volume,and mix.Transfer 10.0mLof this solution to a 50-mLcolor comparison tube,add 3mLof 2Nnitric acid and 2mLof silver nitrate TS,dilute with water to 50mL,and mix.Allow to stand,protected from direct sunlight,for 5minutes.The sample shows no more turbidity than that produced in a solution containing 0.35mLof 0.020Nhydrochloric acid:not more than 0.50%of chloride is found.
Arsenic,Method IIá211ñ:
4ppm.
Heavy metals,Method IIá231ñ:
0.002%.
Limit of pyridine and 2-methylpyridine—
Internal standard solution—
Transfer 1.0mLof 3-methylpyridine to a 50-mLvolumetric flask,dilute with chloroform to volume,and mix.Transfer 1.0mLof this solution to a 50-mLvolumetric flask,dilute with chloroform to volume,and mix.
Standard stock solution—
Transfer about 0.5g each of 2-methylpyridine and pyridine to a 50-mLvolumetric flask,dissolve in chloroform,dilute with chloroform to volume,and mix.Quantitatively dilute 5.0mLof this solution with chloroform to 50.0mL.Transfer 5.0mLof this solution to a 50.0mLvolumetric flask,dilute with chloroform to volume,and mix.
Standard solution—
Transfer 5.0mLofStandard stock solution to a 20-mLvolumetric flask,add 1.0mLof theInternal standard solution,dilute with chloroform to volume,and mix.
Test solution—
Sonicate about 1g of Sucralfate,accurately weighed,in 10.0mLof 1Msodium hydroxide until a uniformly cloudy mixture is obtained.Extract this solution with three 5-mLportions of chloroform,and collect the chloroform extracts in a 20-mLvolumetric flask.Add 1.0mLof theInternal standard solution,dilute with chloroform to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The gas chromatograph is equipped with a flame-ionization detector,a split injection system,and a 0.53-mm ×10-m capillary column coated with a 2.65-µm layer of phase G27.Helium is used as the carrier gas,at a pressure of 36-mm of mercury.The column temperature is maintained at 50 .The injection port temperature and the detector temperature are maintained at 150
.The injection port temperature and the detector temperature are maintained at 150 and 200
and 200 ,respectively.Chromatograph theStandard solution,and record the peak responses as directed forProcedure:the relative retention times of pyridine,2-methylpyridine,and 3-methylpyridine are about 0.42,0.72,and 1.0,respectively;the resolution,R,between pyridine and 2-methylpyridine is not less than 3.5;the resolution,R,between 2-methylpyridine and 3-methylpyridine is not less than 2.5;and the relative standard deviation for replicate injections is not more than 2.0%.
,respectively.Chromatograph theStandard solution,and record the peak responses as directed forProcedure:the relative retention times of pyridine,2-methylpyridine,and 3-methylpyridine are about 0.42,0.72,and 1.0,respectively;the resolution,R,between pyridine and 2-methylpyridine is not less than 3.5;the resolution,R,between 2-methylpyridine and 3-methylpyridine is not less than 2.5;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 1µL)of theStandard solution and theTest solution into the chromatograph,record the chromatograms,and measure the peak responses.Separately calculate the quantities,in µg,of pyridine and 2-methylpyridine,if present,in the portion of Sucralfate taken by the formula:
20C(RU/RS),
in whichCis the concentration,in µg per mL,of pyridine or 2-methylpyridine in theStandard solution;andRUandRSare the peak response ratios of the analyte to the internal standard obtained from theTest solution and theStandard solution,respectively:not more than 0.05%each of pyridine and 2-methylpyridine is found.
Limit of sucrose heptasulfate—
Mobile phase—
Dissolve 99.1g of ammonium sulfate in 900mLof water,dilute with water to 1000mL,and mix.Adjust with phosphoric acid to a pHof 3.5±0.1,filter,and degas.Make adjustments if necessary (seeSystem Suitability underChromatography á621ñ).
Standard solution—
Prepare as directed for theStandard preparation in theAssay.
Chromatographic system—
Prepare as directed in theAssay.
Test solution—
Prepare as directed forAssay preparation in theAssay.
Procedure—
Inject about 50µLof theTest solution into the chromatograph,record the chromatograms,and measure the responses for the major peaks.The relative retention times are about 0.6for sucrose heptasulfate and 1.0for sucrose octasulfate.The ratio of the peak response of the sucrose heptasulfate peak to that of the sucrose octasulfate peak is not more than 0.1.
Aluminum content—
Transfer about 1.0g,accurately weighed,to a 250-mLvolumetric flask,add 10mLof 6.0Nhydrochloric acid,mix,and heat with continuous stirring in a water bath at 70 for 5minutes.Cool to room temperature,dilute with water to volume,and mix.Filter the solution,discarding the first portion of the filtrate.Transfer 25.0mLof the filtrate to a 250-mLbeaker,add 25.0mLof 0.05Medetate disodium VS,add 20mLof acetic acid-ammonium acetate buffer TS,and mix.Heat in a water bath at 70
for 5minutes.Cool to room temperature,dilute with water to volume,and mix.Filter the solution,discarding the first portion of the filtrate.Transfer 25.0mLof the filtrate to a 250-mLbeaker,add 25.0mLof 0.05Medetate disodium VS,add 20mLof acetic acid-ammonium acetate buffer TS,and mix.Heat in a water bath at 70 for 5minutes.Cool to room temperature,add 50mLof alcohol and 2mLof dithizone TS,and mix.Titrate with 0.05Mzinc sulfate VSto a bright rose-pink color.Perform a blank determination and make any necessary correction.Each mLof 0.05Medetate disodium consumed is equivalent to 1.349mg of aluminum:between 15.5%and 18.5%of aluminum is found,calculated on an “as is”basis.
for 5minutes.Cool to room temperature,add 50mLof alcohol and 2mLof dithizone TS,and mix.Titrate with 0.05Mzinc sulfate VSto a bright rose-pink color.Perform a blank determination and make any necessary correction.Each mLof 0.05Medetate disodium consumed is equivalent to 1.349mg of aluminum:between 15.5%and 18.5%of aluminum is found,calculated on an “as is”basis.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Mobile phase—
Dissolve 132g of ammonium sulfate in 900mLof water,dilute with water to 1000mL,and mix.Adjust with phosphoric acid to a pHof 3.5±0.1,filter,and degas.Make adjustments if necessary (seeSystem SuitabilityunderChromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Potassium Sucrose Octasulfate RSinMobile phase,and dilute quantitatively,and stepwise if necessary,withMobile phase to obtain a solution having a known concentration of about 10mg of anhydrous potassium sucrose octasulfate (as determined from the concentration of USP Potassium Sucrose Octasulfate RScorrected for water content by a titrimetric water determination)per mL.
Assay preparation—
Transfer about 450mg of Sucralfate,accurately weighed,to a 35-mLcentrifuge tube,and shake at a moderate rate on a vortex mixer.While shaking add 10.0mLof a mixture of 4.0Nsulfuric acid and 2.2Nsodium hydroxide (1:1).Sonicate with swirling for 5minutes,keeping the temperature of the mixture below 30 .Without delay transfer the tube to a vortex mixer and while shaking at moderate rate,add an accurately measured volume,V,in mL,of 0.1Nsodium hydroxide to bring the pHof the solution to approximately 2,and dilute the solution with (15.0-V)mLof water.Shake for 1minute,and centrifuge for 5minutes.Separate the clear supernatant layer,and allow it to stand at room temperature until the pHstabilizes.If the pHis not between 2.3and 3.5,repeat the test using a different volume of 0.1Nsodium hydroxide.Use the clear supernatant layer.
.Without delay transfer the tube to a vortex mixer and while shaking at moderate rate,add an accurately measured volume,V,in mL,of 0.1Nsodium hydroxide to bring the pHof the solution to approximately 2,and dilute the solution with (15.0-V)mLof water.Shake for 1minute,and centrifuge for 5minutes.Separate the clear supernatant layer,and allow it to stand at room temperature until the pHstabilizes.If the pHis not between 2.3and 3.5,repeat the test using a different volume of 0.1Nsodium hydroxide.Use the clear supernatant layer.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a refractive index detector and a 3.9-mm ×30-cm column that contains packing L8.The detector and column temperatures are maintained at 30 .The flow rate is about 1mLper minute.Chromatograph theStandard preparation,and record the peak responses as directed forProcedure:the column efficiency determined from the sucrose octasulfate peak is not less than 400theoretical plates;the tailing factor for the sucrose octasulfate peak is not more than 4.0;and the relative standard deviation for replicate injections is not more than 2.0%.
.The flow rate is about 1mLper minute.Chromatograph theStandard preparation,and record the peak responses as directed forProcedure:the column efficiency determined from the sucrose octasulfate peak is not less than 400theoretical plates;the tailing factor for the sucrose octasulfate peak is not more than 4.0;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 50µL)of theStandard preparation and theAssay preparation into the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in mg,of sucrose octasulfate (C12H14O35S8)in the portion of Sucralfate taken by the formula:
(974.75/1287.53)(25C)(rU/rS),
in which 974.75and 1287.53are the molecular weights of sucrose octasulfate and anhydrous potassium sucrose octasulfate,respectively;Cis the concentration,in mg per mL,of anhydrous potassium sucrose octasulfate in theStandard preparation;andrUandrSare the peak responses of sucrose octasulfate obtained from theAssay preparation and theStandard preparation,respectively.
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(PA4)Pharmaceutical Analysis 4
USP28–NF23Page 1809
Pharmacopeial Forum:Volume No.28(2)Page 367
Phone Number:1-301-816-8251