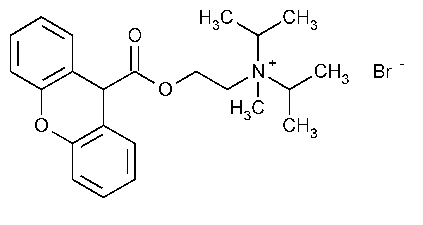
Propantheline Bromide
2-Propanaminium,N-methyl-N-(1-methylethyl)-N-[2-[(9H-xanthen-9-ylcarbonyl)oxy]ethyl]-,bromide.
(2-Hydroxyethyl)diisopropylmethylammonium bromide xanthene-9-carboxylate [50-34-0].
»Propantheline Bromide contains not less than 98.0percent and not more than 102.0percent of C23H30BrNO3,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
USP Reference standards á11ñ—
USP Xanthanoic Acid RS.USP Xanthone RS.USP Propantheline Bromide RS.USP Propantheline Bromide Related Compound A RS.
Identification—
A:
Prepare 3mLof a solution in chloroform having a concentration of about 6mg per mL,and reserve a 1-mLportion for Identificationtest B.In a well-ventilated hood,apply 2mLof this solution dropwise to a salt plate while continuously evaporating the solvent with the aid of an IRheat lamp and a current of dry air.Heat the residue at 105 for 15minutes:the IRabsorption spectrum of the residue on the single salt plate exhibits maxima only at the same wavelengths as that of a similar preparation of USP Propantheline Bromide RS,treated in the same manner.
for 15minutes:the IRabsorption spectrum of the residue on the single salt plate exhibits maxima only at the same wavelengths as that of a similar preparation of USP Propantheline Bromide RS,treated in the same manner.
B:
Apply 5µLof the chloroform solution retained from Identificationtest Aand 5µLof a Standard solution of USP Propantheline Bromide RSin chloroform containing 6mg per mLto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel.Develop the chromatogram in a solvent system consisting of a mixture of 1Nhydrochloric acid and acetone (1:1)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the developing chamber,mark the solvent front,and dry at 105 for 5minutes.Spray the plate with potassium-bismuth iodide TS,and heat at 105
for 5minutes.Spray the plate with potassium-bismuth iodide TS,and heat at 105 for 5minutes:the RFvalue of the principal spot obtained from the test solution corresponds to that obtained from the Standard solution.
for 5minutes:the RFvalue of the principal spot obtained from the test solution corresponds to that obtained from the Standard solution.
C:
To 5mLof a solution (1in 100)add 2mLof 2Nnitric acid:this solution responds to the tests for Bromide á191ñ,except that in the test that liberates bromine,the chloroform layer may be yellow.
Loss on drying á731ñ—
Dry it at 105 for 4hours:it loses not more than 0.5%of its weight.
for 4hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Related compounds—
pH3.5buffer solution—
Dissolve 17.3g of sodium dodecyl sulfate in 1000mLof water containing 10mLof phosphoric acid in a 2000-mLvolumetric flask.Add 250mLof 0.5Msodium hydroxide and,while stirring,adjust with 0.5Msodium hydroxide or dilute phosphoric acid (1in 10)to a pHof 3.5±0.05,dilute with water to volume,and mix.
Mobile phase—
Prepare a filtered and degassed mixture of acetonitrile andpH3.5buffer solution(55:45).Make adjustments if necessary (seeSystem SuitabilityunderChromatography á621ñ).
Standard solution—
Dissolve accurately weighed quantities of USP Propantheline Bromide Related Compound A RS,USP Xanthanoic Acid RS,and USP Xanthone RSinMobile phase,and dilute quantitatively,and stepwise if necessary,withMobile phaseto obtain a solution having a known concentration of about 6.0µg of propantheline bromide related compound Aper mL,and about 1.5µg each of xanthanoic acid and xanthone per mL.
Test solution—
Transfer about 60mg of Propantheline Bromide,accurately weighed,to a 200-mLvolumetric flask,dissolve inMobile phase,dilute withMobile phaseto volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 254-nm detector and a 4.6-mm ×25-cm column that contains packing L7.The flow rate is about 2.0mLper minute.Chromatograph theStandard solution,and record peak responses as directed forProcedure:the resolution,R,between the least resolved peaks is not less than 1.2;and the relative standard deviation for replicate injections of theStandard solutionis not more than 6.0%for each component.
Procedure—
Separately inject equal volumes (about 50µL)of theStandard solutionand theTest solutioninto the chromatograph,record the chromatograms for a total time of not less than 1.5times the retention time of the propantheline bromide peak,and measure the response for each peak,except the peaks at or before the void volume.Calculate the percentage of xanthanoic acid,xanthone,and propantheline bromide related compound Agreater than or equal to 0.1%in the portion of Propantheline Bromide taken by the formula:
20C/W(rU/rS),
in whichCis the concentration,in µg,of xanthanoic acid,xanthone,or propantheline bromide related compound Aper mLof theStandard solution;Wis the weight,in mg,of Propantheline Bromide taken;andrUandrSare the related compound peak responses obtained from theTest solutionand theStandard solution,respectively:not more than 2.0%of propantheline bromide related compound Aand 0.5%each of xanthone and xanthanoic acid is found.Calculate the percentage of all unknown impurities greater than or equal to 0.1%by the formula:
100ri/rt,
in whichriis the response of the unknown impurity peak;andrtis the sum of the responses of all the measured peaks observed in the chromatogram:the sum total of all known and unknown impurities is not more than 3.0%.
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Bromide content—
Weigh accurately about 500mg,and dissolve in 40mLof water.Add 10mLof glacial acetic acid and 40mLof methanol,then add eosin Y TS,and titrate with 0.1Nsilver nitrate VS.Each mLof 0.1Nsilver nitrate is equivalent to 7.990mg of Br.Not less than 17.5%and not more than 18.2%of Br,calculated on the dried basis,is found.
Assay—
Dissolve about 600mg of Propantheline Bromide,accurately weighed,in a mixture of 20mLof glacial acetic acid and 15mLof mercuric acetate TS,warming slightly if necessary to effect solution.Cool to room temperature,and titrate with 0.1Nperchloric acid VS,determining the endpoint potentiometrically.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 44.84mg of C23H30BrNO3.
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(PA4)Pharmaceutical Analysis 4
USP28–NF23Page 1644
Pharmacopeial Forum:Volume No.29(2)Page 430
Phone Number:1-301-816-8251