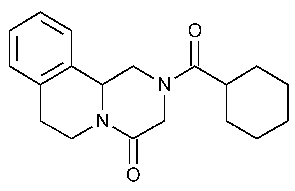
Praziquantel
4H-Pyrazino[2,1-a]isoquinolin-4-one,2-(cyclohexylcarbonyl)-1,2,3,6,7,11b-hexahydro-.
2-(Cyclohexylcarbonyl)-1,2,3,6,7,11b-hexahydro-4H-pyrazino-[2,1-a]isoquinolin-4-one [55268-74-1].
»Praziquantel contains not less than 98.5percent and not more than 101.0percent of C19H24N2O2,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed,light-resistant containers.
USP Reference standards á11ñ—
USP Praziquantel RS.USP Praziquantel Related Compound A RS.USP Praziquantel Related Compound B RS.USP Praziquantel Related Compound C RS.
Identification,
Infrared Absorption á197Kñ.
Loss on drying á731ñ—
Dry it in vacuum at a pressure not exceeding 5mm of mercury at 50 over phosphorus pentoxide for 2hours:it loses not more than 0.5%of its weight.
over phosphorus pentoxide for 2hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Phosphate—
Cupric sulfate solution—
Dissolve 250mg of cupric sulfate and 4.5g of ammonium acetate in sufficient 2Nacetic acid to obtain 100mLof solution.
4-Amino-3-hydroxy-1-naphthalenesulfonic acid solution—
Grind in a mortar 5g of sodium sulfite,94.3g of sodium metabisulfite,and 700mg of 4-amino-3-hydroxy-1-naphthalenesulfonic acid.Dissolve 1.5g of this mixture in 10mLof water,heating gently if necessary.Use this solution only when freshly prepared.
Standard solution—
Dissolve 143.3mg of dried monobasic potassium phosphate in water to make 1000mLof solution.Transfer 5.0mLof this solution to a 100-mLvolumetric flask,dilute with water to volume,and mix.This solution contains the equivalent of 5µg of phosphate (PO4)in each mL.
Test solution—
Add 30mLof water to 500mg of specimen,and heat to boiling.Allow to cool,and filter,collecting the filtrate in a 50-mLvolumetric flask.Wash the filter with water,collecting the washings in the volumetric flask,dilute with water to volume,and mix.
Procedure—
Treat 10mLofTest solution and 10mLofStandard solution as follows.To each add 5mLofCupric sulfate solution,2mLof ammonium molybdate solution (3in 100),1mLof4-Amino-3-hydroxy-1-naphthalenesulfonic acid solution,and 1mLof perchloric acid solution (3in 100),mix,and allow to stand for 15minutes:theTest solution does not have a blue color that is darker than that of theStandard solution (0.05%).
Heavy metals,Method IIá231ñ:
0.002%.
Related compounds—
Mobile phaseand Chromatographic system—
Prepare as directed in theAssay.
Standard preparation—
Dissolve accurately weighed quantities of USP Praziquantel Related Compound A RS,USP Praziquantel Related Compound B RS,and USP Praziquantel Related Compound C RSinMobile phase to obtain a single solution having known concentrations of about 0.04mg of each per mL.
Test preparation—
Transfer about 200mg of Praziquantel,accurately weighed,to a 10-mLvolumetric flask,dissolve in and dilute withMobile phase to volume,and mix.
Procedure—
Separately inject equal volumes (about 10µL)of theStandard preparation and theTest preparation into the chromatograph,record the chromatograms,and measure the responses for the peaks.The relative retention times are about 0.8for praziquantel related compound A,1.0for praziquantel,1.8for praziquantel related compound B,and 2.1for praziquantel related compound C.Calculate,in turn,the percentages of 2-benzoyl-1,2,3,6,7,11b-hexahydro-4H-pyrazino[2,1-a]isoquinolin-4-one (praziquantel related compound A),2-(cyclohexylcarbonyl)-2,3,6,7-tetrahydro-4H-pyrazino[2,1-a]isoquinolin-4-one (praziquantel related compound B),and 2-(N-formylhexahydrohippuroyl)-1,2,3,4-tetrahydroisoquinolin-1-one (praziquantel related compound C)in the portion of Praziquantel taken by the formula:
1000(C/W)(rU/rS),
in whichCis the concentration,in mg per mL,of the respective USP Reference Standard taken to prepare theStandard preparation;Wis the weight,in mg,of Praziquantel taken to prepare theTest preparation;andrUandrSare the peak responses at corresponding retention times,obtained from theTest preparation and theStandard preparation,respectively.Not more than 0.2%of each is found.
Assay—
Mobile phase—
Prepare a suitable degassed mixture of acetonitrile and water (60:40).Make adjustments if necessary (seeSystem SuitabilityunderChromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Praziquantel RSinMobile phase,and dilute quantitatively,and stepwise if necessary,withMobile phase to obtain a solution having a known concentration of about 0.18mg per mL.
Assay preparation—
Transfer about 36mg of Praziquantel,accurately weighed,to a 20-mLvolumetric flask,dissolve in and dilute withMobile phase to volume,and mix.Transfer 5.0mLof the solution so obtained to a 50-mLvolumetric flask,dilute withMobile phase to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 210-nm detector and a 4-mm ×25-cm column that contains 10-µm packing L1.The flow rate is about 1.5mLper minute.Chromatograph theStandard preparation,and record the peak responses as directed forProcedure:the tailing factor is not more than 1.5;and the relative standard deviation for replicate injections is not more than 1.0%.
Procedure—
Separately inject equal volumes (about 10µL)of theStandard preparation and theAssay preparation into the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in mg,of C19H24N2O2in the portion of Praziquantel taken by the formula:
200C(rU/rS),
in whichCis the concentration,in mg per mL,ofUSP Praziquantel RSin theStandard preparation;andrUandrSare the peak responses obtained from theAssay preparation and theStandard preparation,respectively.
Auxiliary Information—
Staff Liaison:Behnam Davani,Ph.D.,MBA,Senior Scientist
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 1605
Pharmacopeial Forum:Volume No.28(6)Page 1853
Phone Number:1-301-816-8394