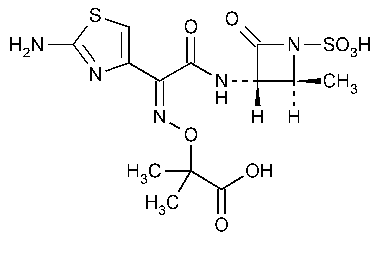
Aztreonam
Propanoic acid,2-[[[1-(2-amino-4-thiazolyl)-2-[(2-methyl-4-oxo-1-sulfo-3-azetidinyl)amino]-2-oxoethylidene]amino]oxy]-2-methyl-,[2S-[2a,3b(Z)]]-.
(Z)-2-[[[(2-Amino-4-thiazolyl)[[(2S,3S)-2-methyl-4-oxo-1-sulfo-3-azetidinyl]carbamoyl]methylene]amino]oxy]-2-methylpropionic acid [78110-38-0].
»Aztreonam contains not less than 90.0percent and not more than 105.0percent of C13H17N5O8S2.
Packaging and storage—
Preserve in tight containers.
Labeling—
Where it is intended for use in preparing injectable dosage forms,the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
Change to read:
Identification,Infrared Absorption á197Kñ—
Bacterial endotoxins á85ñ—
Where the label states that aztreonam is sterile or must be subjected to further processing during the preparation of injectable dosage forms,it contains not more than 0.17USP Endotoxin Unit per mg of aztreonam.
Sterility á71ñ—
Where the label states that Aztreonam is sterile,it meets the requirements when tested as directed for Membrane Filtrationunder Test for Sterility of the Product to be Examined,using Fluid Ato which 23.4g of sterile arginine has been added to each 1000mL.
Water,Method Iá921ñ:
not more than 2.0%.
Residue on ignition á281ñ:
not more than 0.1%,the charred residue being moistened with 2mLof nitric acid and 5drops of sulfuric acid.
Heavy metals,Method IIá231ñ:
0.003%.
Assay—
Mobile phase—
Dissolve 6.8g of monobasic potassium phosphate in water to make 1000mL,and adjust with 1Mphosphoric acid to a pHof 3.0±0.1.Prepare a suitable mixture of this solution and methanol (4:1).Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Quantitatively dissolve an accurately weighed quantity of USP Aztreonam RSin Mobile phaseto obtain a solution having a known concentration of about 1mg per mL.
Resolution solution—
Prepare a solution in Mobile phasecontaining in each mLabout 0.2mg each of USP Aztreonam RSand USP Aztreonam E-Isomer RS.
Assay preparation—
Transfer about 25mg of Aztreonam,accurately weighed,to a 25-mLvolumetric flask,dissolve in and dilute with Mobile phaseto volume,and mix.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 270-nm detector,a 2-mm ×10-cm precolumn containing packing L2,and a 4.6-mm ×30-cm analytical column containing packing L1.The flow rate is about 1.5mLper minute.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the relative retention times are about 0.6for aztreonam and 1.0for aztreonam E-isomer;the resolution,R,between aztreonam and aztreonam E-isomer is not less than 2.0;the column efficiency as determined from the aztreonam peak is not less than 1000theoretical plates;the tailing factor for the aztreonam peak is not more than 2.0;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 20µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the percentage of C13H17N5O8S2in the portion of Aztreonam taken by the formula:
2.5(CSPS/W)(rU/rS),
in which CSis the concentration,in mg per mL,of USP Aztreonam RSin the Standard preparation;PSis the assigned purity,in µg per mg,of USP Aztreonam RS;Wis the weight,in mg,of Aztreonam taken to prepare the Assay preparation;and rUand rSare the aztreonam peak responses obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 210
Pharmacopeial Forum:Volume No.30(1)Page 61
Phone Number:1-301-816-8335