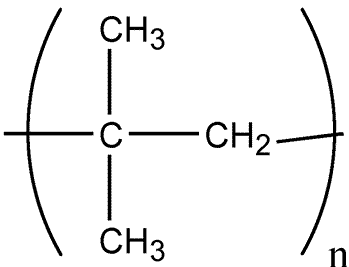
Polyisobutylene
»Polyisobutylene is a synthetic polymer produced by the low-temperature polymerization of isobutylene in liquid ethylene,methylene chloride,or hexane,using an aluminum-chloride or boron-trifluoride catalyst.
Packaging and storage—
Store in well-closed containers.Store at room temperature,and avoid excessive heat and freezing.Protect from moisture.
Labeling—
Label it to indicate the nominal molecular weight and the solvent used in the polymerization process.
USP Reference standards á11ñ—
USP Polyisobutylene RS.
Identification,Infrared Absorption á197Fñ—
Prepare the sample by dissolving it in hot toluene and evaporating on a salt plate.
Loss on drying á731ñ—
Dry a 5-g sample for 2hours at 105 :it loses not more than 0.1%of its weight.
:it loses not more than 0.1%of its weight.
Lead á251ñ—
Accurately weigh about 3.3g of Polyisobutylene,transfer to a porcelain dish,and heat on a hot plate until completely charred.Then heat in a muffle furnace at 480 for 8hours,and cool.Cautiously add 5mLof nitric acid,evaporate to dryness on a hot plate,then heat again in the muffle furnace for exactly 15minutes,and cool.Extract the ash with two 10-mLportions of water,filtering each extract into a separator.Leach any insoluble material on the filter with 6mLof Ammonium Citrate Solution,2mLof Hydroxylamine Hydrochloride Solution,and 5mLof water,adding the filtered washings to the separator.Continue as directed under Procedure,beginning with “Add two drops of phenol red TS”,and use 10mLof Diluted Standard Lead Solution(10µg of lead)for the control:the color of the sample solution does not exceed that in the control (0.0003%).
for 8hours,and cool.Cautiously add 5mLof nitric acid,evaporate to dryness on a hot plate,then heat again in the muffle furnace for exactly 15minutes,and cool.Extract the ash with two 10-mLportions of water,filtering each extract into a separator.Leach any insoluble material on the filter with 6mLof Ammonium Citrate Solution,2mLof Hydroxylamine Hydrochloride Solution,and 5mLof water,adding the filtered washings to the separator.Continue as directed under Procedure,beginning with “Add two drops of phenol red TS”,and use 10mLof Diluted Standard Lead Solution(10µg of lead)for the control:the color of the sample solution does not exceed that in the control (0.0003%).
Molecular weight—
Solvent—
Use diisobutylene.
Test solution 1—
Dissolve 1g of Polyisobutylene,accurately weighed,in 95mLof Solvent,filter into a 100-mLvolumetric flask,dilute with Solvent to volume,and mix.Transfer 50.0mLof the solution so obtained into a tared dish,evaporate on a steam bath for about 1hour,and then complete the evaporation to dryness by heating in the vacuum oven at 70 to constant weight.Calculate the concentration of Test solution 1.
to constant weight.Calculate the concentration of Test solution 1.
Test solution 2—
Transfer 2.0mLof Test solution 1to a 50-mLvolumetric flask,and dilute with Solventto volume.Calculate the concentration of Test solution 2.
Test solution 3—
Transfer 5.0mLof Test solution 1to a 50-mLvolumetric flask,and dilute with Solventto volume.Calculate the concentration of Test solution 3.
Test solution 4—
Transfer 10.0mLofTest solution 1to a 50-mLvolumetric flask,and dilute with Solventto volume.Calculate the concentration of Test solution 4.
Procedure—
Using a Cannon-Fenske,or equivalent viscosimeter,having dimensions such that the flow time is not less than 200seconds,measure the flow of the Solvent and of each of the four Test solutionsat 20 .Calculate the specific viscosity,hsp,of Test solution 1by the formula:
.Calculate the specific viscosity,hsp,of Test solution 1by the formula:
(t/t0)–1,
in whicht0is the flow time,in seconds,of the Solvent;and tis the flow time,in seconds,of Test solution 1.Calculate the reduced viscosity of Test solution 1by the formula:
hsp /C,
in which hspis the specific viscosity,calculated above;and Cis the concentration,in g per 100mL,of Test solution 1.Calculate the specific viscosity and the reduced viscosity of Test solutions 2,3,and 4in the same manner as Test solution 1.
Plot the reduced viscosity of each solution against concentration,and extrapolate to zero concentration to obtain the intrinsic viscosity,[h].Finally,calculate the molecular weight of Polyisobutylene by the formula:
([h]/K)1/a,
in which Kis 3.60×10–4and ais 0.64.
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(EMC)Excipients:Monograph Content
USP28–NF23Page 3058
Pharmacopeial Forum:Volume No.30(3)Page 990
Phone Number:1-301-816-8251