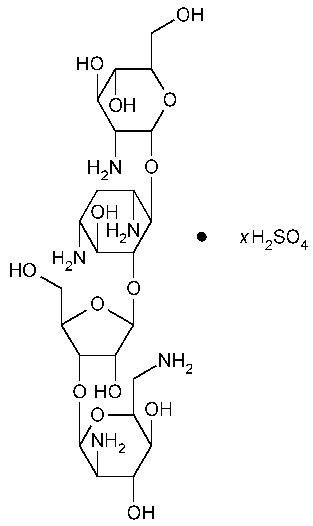
Paromomycin Sulfate
D-Streptamine,[O-2-amino-2-deoxy-a-D-glucopyranosyl-(1®4)-O-[O-2,6-diamino-2,6-dideoxy-b-L-idopyranosyl-(1®3)-b-D-ribofuranosyl-(1®5)]-2-deoxy-,sulfate (salt).
O-2,6-Diamino-2,6-dideoxy-b-L-idopyranosyl-(1®3)-O-b-D-ribofuranosyl-(1®5)-O-[2-amino-2-deoxy-a-D-glucopyranosyl-(1®4)]-2-deoxystreptamine sulfate (salt) [1263-89-4].
Base 615.64 [59-04-1;7542-37-2].
»Paromomycin Sulfate is the sulfate salt of an antibiotic substance or substances produced by the growth of Streptomyces rimosusvar.paromomycinus,or a mixture of two or more such salts.It has a potency equivalent to not less than 675µg of paromomycin (C23H45N5O14)per mg,calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
A:
Prepare a test solution in water containing 10mg of paromomycin per mL.Apply 25µLof this solution and 25µLof a Standard solution of USP Paromomycin Sulfate RScontaining 10mg of paromomycin per mLto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel.Allow the spots to dry,place the plate in a developing chamber,and develop the chromatogram in a solvent system consisting of a mixture of freshly prepared ammonium acetate solution (4in 100),n-propyl alcohol,and ammonium hydroxide (30:10:6)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the chamber,mark the solvent front,and allow it to air-dry for 10minutes.Heat the plate at 105 for 1hour,allow to cool,and spray with a solution of ninhydrin in butanol (1in 100).Heat the plate at 105
for 1hour,allow to cool,and spray with a solution of ninhydrin in butanol (1in 100).Heat the plate at 105 for 5minutes:paromomycin appears as a red spot,and the RFvalue of the principal spot obtained from the test solution corresponds to that obtained from the Standard solution.
for 5minutes:paromomycin appears as a red spot,and the RFvalue of the principal spot obtained from the test solution corresponds to that obtained from the Standard solution.
B:
It meets the requirements of the tests for Sulfate á191ñ.
Specific rotation á781Sñ:
between +50 and +55
and +55 .
.
Test solution:
50mg per mL,in water.
pHá791ñ:
between 5.0and 7.5,in a solution (3in 100).
Loss on drying á731ñ—
Dry about 100mg in a capillary-stoppered bottle in vacuum at a pressure not exceeding 5mm of mercury at 60 for 3hours:it loses not more than 5.0%of its weight.
for 3hours:it loses not more than 5.0%of its weight.
Residue on ignition á281ñ:
not more than 2.0%,the charred residue being moistened with 2mLof nitric acid and 5drops of sulfuric acid.
Assay—
Proceed with Paromomycin Sulfate as directed under Antibiotics—Microbial Assays á81ñ.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 1473
Phone Number:1-301-816-8335