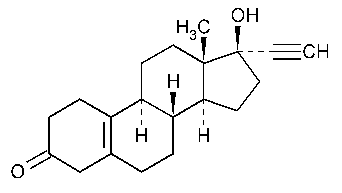
Norethynodrel
19-Norpregn-5(10)-en-20-yn-3-one,17-hydroxy-,(17a)-.
17-Hydroxy-19-nor-17a-pregn-5(10)-en-20-yn-3-one [68-23-5].
»Norethynodrel contains not less than 97.0percent and not more than 101.0percent of C20H26O2.
Packaging and storage—
Preserve in well-closed containers.
Identification,Infrared Absorption á197Sñ—
Solution:
1in 20.
Medium:
chloroform.
Specific rotation á781Sñ:
between +119 and +125
and +125 .
.
Test solution:
10mg per mL,in dioxane.
Limit of ethynyl group—
Dissolve 200mg in about 40mLof tetrahydrofuran.Add 10mLof silver nitrate solution (1in 10),and titrate with 0.1Nsodium hydroxide VS,using either a glass-calomel or a silver-silver chloride electrode system with potassium nitrate filling solution.Each mLof 0.1Nsodium hydroxide is equivalent to 2.503mg of ethynyl group (–CºCH).Not less than 8.18%and not more than 8.43%of ethynyl group is found.
Limit of norethindrone—
Test preparation—
Prepare a solution of Norethynodrel in chloroform containing 10mg per mL.
Standard solution—
Prepare a solution of USP Norethindrone RSin chloroform to contain 1mg per mL.Dilute 2mLof the solution with chloroform to 10mL.
Procedure—
Apply 10-µLvolumes of the Test preparationand the Standard solution(see Chromatography á621ñ)to a thin-layer chromatographic plate coated with a 0.25-mm layer of chromatographic silica gel mixture,and allow not more than 5minutes between spotting the plate and starting development of the chromatogram.Place the plate in a suitable chromatographic chamber previously equilibrated with a mixture of cyclohexane,ethyl acetate,and methanol (60:40:2),and allow the solvent front to move 15cm.Spray the plate with dilute sulfuric acid (1in 2),heat the plate at 105 for 5minutes,and view under long-wavelength UVlight.Locate any norethindrone impurity in the Test preparationby comparison with the RFvalue from the Standard solution.If present,the norethindrone spot from the Test preparationis not larger or more intense than the spot from the Standard solution(2.0%).
for 5minutes,and view under long-wavelength UVlight.Locate any norethindrone impurity in the Test preparationby comparison with the RFvalue from the Standard solution.If present,the norethindrone spot from the Test preparationis not larger or more intense than the spot from the Standard solution(2.0%).
Ordinary impurities á466ñ—
Test solution:
chloroform.
Standard solution:
chloroform.
Eluant:
ether.
Visualization:
5,followed by viewing under long-wavelength UVlight.
Organic volatile impurities,Method Vá467ñ:
meets the requirements.
Solvent—
Use dimethyl sulfoxide.
Assay—
Standard preparation—
Dissolve a suitable quantity of USP Norethynodrel RS,accurately weighed,in methanol,and dilute quantitatively with methanol to obtain a solution having a known concentration of about 1mg per mL.
Assay preparation—
Dissolve about 100mg of Norethynodrel,accurately weighed,in methanol to make 100.0mL,and mix.
Procedure—
Transfer 10.0mLeach of the Standard preparationand the Assay preparationto separate 100-mLvolumetric flasks.To each flask add 40mLof methanol,then add 5mLof a mixture of 3volumes of hydrochloric acid and 2volumes of water,mix quickly,and allow to stand at a temperature of about 25 for 1hour,accurately timed.Prior to the end of the 1-hour period,prepare blanks as follows.Add 1.0mLeach of the Standard preparationand the Assay preparationto separate 100-mLvolumetric flasks,each containing a mixture of 50mLof methanol and 2mLof water,dilute each with methanol to volume,and mix.At the end of the 1-hour reaction period,dilute each of the acid-containing solutions with methanol to volume,and mix.Transfer 10.0mLof each into separate 100-mLvolumetric flasks,add 2mLof water to each,dilute with methanol to volume,and mix.Concomitantly determine the absorbances of the solutions in 1-cm cells at the wavelength of maximum absorbance at about 240nm,with a suitable spectrophotometer,relative to the corresponding blanks.Calculate the quantity,in mg,of C20H26O2in the portion of Norethynodrel taken by the formula:
for 1hour,accurately timed.Prior to the end of the 1-hour period,prepare blanks as follows.Add 1.0mLeach of the Standard preparationand the Assay preparationto separate 100-mLvolumetric flasks,each containing a mixture of 50mLof methanol and 2mLof water,dilute each with methanol to volume,and mix.At the end of the 1-hour reaction period,dilute each of the acid-containing solutions with methanol to volume,and mix.Transfer 10.0mLof each into separate 100-mLvolumetric flasks,add 2mLof water to each,dilute with methanol to volume,and mix.Concomitantly determine the absorbances of the solutions in 1-cm cells at the wavelength of maximum absorbance at about 240nm,with a suitable spectrophotometer,relative to the corresponding blanks.Calculate the quantity,in mg,of C20H26O2in the portion of Norethynodrel taken by the formula:
100C(AU/AS),
in which Cis the concentration,in mg per mL,of USP Norethynodrel RSin the Standard preparation,and AUand ASare the absorbances of the solutions from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Clydewyn M.Anthony,Ph.D.,Scientist
Expert Committee:(PA1)Pharmaceutical Analysis 1
USP28–NF23Page 1398
Phone Number:1-301-816-8139