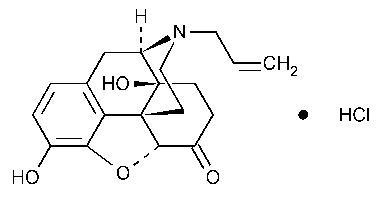
Naloxone Hydrochloride
C19H21NO4·HCl
363.84
Morphinan-6-one,4,5-epoxy-3,14-dihydroxy-17-(2-propenyl)-,hydrochloride,(5a)-.
17-Allyl-4,5a-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride [357-08-4].
Morphinan-6-one,4,5-epoxy-3,14-dihydroxy-17-(2-propenyl)-,hydrochloride,(5a)-.
17-Allyl-4,5a-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride [357-08-4].
Dihydrate 399.87 [51481-60-8].
»Naloxone Hydrochloride is anhydrous or contains two molecules of water of hydration.It contains not less than 98.0percent and not more than 100.5percent of C19H21NO4·HCl,calculated on the dried basis.
Packaging and storage—
Preserve in tight,light-resistant containers.Store at 25 ,excursions permitted between 15
,excursions permitted between 15 and 30
and 30 .
.
Identification,Infrared Absorption á197Kñ—
Test specimen—
Dissolve about 150mg in 25mLof water in a small separator,add a few drops of 6Nammonium hydroxide slowly until no more white precipitate is formed.Extract with three 5-mLportions of chloroform,pass the extracts through a dry filter,collecting the filtrate in a small flask.Evaporate the filtrate on a steam bath to dryness,and dry the residue at 105 for one hour.
for one hour.
Specific rotation á781Sñ:
between -170 and -181
and -181 .
.
Test solution:
25mg per mL,in water.
Loss on drying á731ñ—
Dry it at 105 to constant weight:the anhydrous form loses not more than 0.5%of its weight,and the hydrous form loses not more than 11.0%of its weight.
to constant weight:the anhydrous form loses not more than 0.5%of its weight,and the hydrous form loses not more than 11.0%of its weight.
Noroxymorphone hydrochloride [(–)-4,5a-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride]and other impurities—
Transfer about 40mg,accurately weighed,to a 5-mLvolumetric flask,dissolve completely in 2.0mLof water,add methanol to volume,and mix,to obtain the test solution.Prepare a solution of USP Naloxone RSin chloroform containing about 7.6mg per mL.Prepare a solution of USP Noroxymorphone Hydrochloride RSin dilute methanol (3in 5)containing 0.084mg per mL.Apply 5µLeach of the test solution and the two Standard solutions to a thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel that previously has been activated by heating for 15minutes at 105 .Immediately place the plate in a suitable chromatographic chamber containing a 1in 20solution of methanol in ammoniacal butanol previously prepared by shaking 100mLof butyl alcohol with 60mLof ammonium hydroxide solution (1in 100)and discarding the lower layer.Develop the chromatogram,protected from light,until the solvent front has moved about 10cm from the point of application.Remove the plate,dry thoroughly,and spray with ferric chloride-potassium ferricyanide reagent prepared,immediately prior to use,by dissolving 100mg of potassium ferricyanide in 20mLof ferric chloride solution (1in 10).Other than the principal spot corresponding inRFvalue to that of USP Naloxone RSand the spot at the origin (ammonium chloride),no other spot is more intense than the spot corresponding to that of USP Noroxymorphone Hydrochloride RS(1.0%).
.Immediately place the plate in a suitable chromatographic chamber containing a 1in 20solution of methanol in ammoniacal butanol previously prepared by shaking 100mLof butyl alcohol with 60mLof ammonium hydroxide solution (1in 100)and discarding the lower layer.Develop the chromatogram,protected from light,until the solvent front has moved about 10cm from the point of application.Remove the plate,dry thoroughly,and spray with ferric chloride-potassium ferricyanide reagent prepared,immediately prior to use,by dissolving 100mg of potassium ferricyanide in 20mLof ferric chloride solution (1in 10).Other than the principal spot corresponding inRFvalue to that of USP Naloxone RSand the spot at the origin (ammonium chloride),no other spot is more intense than the spot corresponding to that of USP Noroxymorphone Hydrochloride RS(1.0%).
Chloride content—
Dissolve about 300mg,accurately weighed,in 50mLof methanol contained in a 125-mLconical flask,add 5mLof glacial acetic acid and 2drops of eosin Y TS,and titrate with 0.1Nsilver nitrate VSto a pink endpoint.Each mLof 0.1Nsilver nitrate is equivalent to 3.545mg of chloride.Not less than 9.54%and not more than 9.94%,calculated on the dried basis,is found.
Assay—
Dissolve about 300mg of Naloxone Hydrochloride,previously dried and accurately weighed,in a mixture of 40mLof glacial acetic acid and 10mLof acetic anhydride;add 10mLof mercuric acetate TSand 1drop of methyl violet TS;and titrate with 0.1Nperchloric acid VS.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 36.38mg of C19H21NO4·HCl.
Auxiliary Information—
Staff Liaison:Daniel K.Bempong,Ph.D.,Scientist
Expert Committee:(PA2)Pharmaceutical Analysis 2
USP28–NF23Page 1328
Pharmacopeial Forum:Volume No.29(6)Page 1936
Phone Number:1-301-816-8143