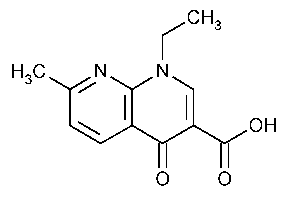
Nalidixic Acid
1,8-Naphthyridine-3-carboxylic acid,1-ethyl-1,4-dihydro-7-methyl-4-oxo-.
1-Ethyl-1,4-dihydro-7-methyl-4-oxo-1,8-naphthyridine-3-carboxylic acid [389-08-2].
»Nalidixic Acid contains not less than 99.0percent and not more than 101.0percent of C12H12N2O3,calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
A:
Infrared Absorption á197Kñ.
B:
Ultraviolet Absorption á197Uñ—
Solution:
5µg per mL.
Medium:
0.01Nsodium hydroxide.
Absorptivities at 258nm,calculated on the dried basis,do not differ by more than 3.0%.
Loss on drying á731ñ—
Dry it at 105 for 2hours:it loses not more than 0.5%of its weight.
for 2hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Heavy metals,Method IIá231ñ:
0.002%.
Chromatographic purity—
Standard preparation—
Prepare a solution of USP Nalidixic Acid RSin chloroform containing 1.0mg per mL.Dilute quantitatively with chloroform to obtain Standard preparationshaving the following composition:
| Standard preparation |
Dilution | Concentration (mg RS per mL) |
Percentage (%, for comparison with test specimen) |
| A | 1in 10 | 0.1 | 0.5 |
| B | 1in 25 | 0.04 | 0.2 |
| C | 1in 50 | 0.02 | 0.1 |
Test preparation—
Dissolve an accurately weighed quantity of Nalidixic Acid in chloroform to obtain a solution containing 20mg per mL.
Procedure—
Apply separately 10µLof the Test preparationand 10µLof each Standard preparationto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel mixture.Position the plate in a chromatographic chamber,and develop the chromatograms in a solvent system consisting of a mixture of alcohol,chloroform,and 5Mammonium hydroxide (70:20:10)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the developing chamber,mark the solvent front,and allow the solvent to evaporate,with the aid of warm circulating air.Examine the plate under short-wavelength UVlight.Compare the intensities of any secondary spots observed in the chromatogram of the Test preparationwith those of the principal spots in the chromatograms of the Standard preparations:no secondary spot is more intense than the principal spot obtained from Standard preparation A(0.5%),and the sum of the intensities of all secondary spots obtained from the Test preparationdoes not exceed 1.0%.
Assay—
Dissolve about 250mg of Nalidixic Acid,accurately weighed,in 30mLof dimethylformamide that previously has been neutralized to thymolphthalein TS,and titrate with 0.1Nlithium methoxide VS,using a magnetic stirrer and taking precautions against absorption of atmospheric carbon dioxide.Each mLof 0.1Nlithium methoxide is equivalent to 23.22mg of C12H12N2O3.
Auxiliary Information—
Staff Liaison:Behnam Davani,Ph.D.,MBA,Senior Scientist
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 1326
Pharmacopeial Forum:Volume No.30(1)Page 132
Phone Number:1-301-816-8394