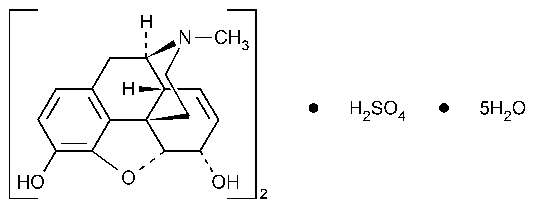
Morphine Sulfate
(C17H19NO3)2·H2SO4·5H2O
758.83
Morphinan-3,6-diol,7,8-didehydro-4,5-epoxy-17-methyl,(5a,6a)-,sulfate (2:1)(salt),pentahydrate.
7,8-Didehydro-4,5a-epoxy-17-methylmorphinan-3,6a-diol sulfate (2:1)(salt)pentahydrate [6211-15-0].
Morphinan-3,6-diol,7,8-didehydro-4,5-epoxy-17-methyl,(5a,6a)-,sulfate (2:1)(salt),pentahydrate.
7,8-Didehydro-4,5a-epoxy-17-methylmorphinan-3,6a-diol sulfate (2:1)(salt)pentahydrate [6211-15-0].
Anhydrous 668.77 [64-31-3].
»Morphine Sulfate contains not less than 98.0percent and not more than 102.0percent of (C17H19NO3)2·H2SO4,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight,light-resistant containers.Store at 25 ,excursions permitted between 15
,excursions permitted between 15 and 30
and 30 .
.
Identification—
A:
Infrared Absorption á197Kñ:dried at 145 for 1hour.
for 1hour.
B:
To 1mg in a porcelain crucible or small dish add 0.5mLof sulfuric acid containing,in each mL,1drop of formaldehyde TS:an intense purple color is produced at once,and quickly changes to deep blue-violet(distinction from codeine,which gives at once an intense violet-blue color,and from hydromorphone,which gives at first a yellow to brown color,changing to pink and then to purplish red).
C:
To a solution of 5mg in 5mLof sulfuric acid in a test tube add 1drop of ferric chloride TS,mix,and heat in boiling water for 2minutes:a blue color is produced,and when 1drop of nitric acid is added,it changes to dark red-brown(codeine and ethylmorphine give the same color reactions,but hydromorphone and papaverine do not produce this color change).
D:
Asolution (1in 50)responds to the tests forSulfate á191ñ.
Specific rotation á781Sñ:
between -107 and -109.5
and -109.5 .
.
Test solution:
the equivalent of 20mg per mL,in water.
Acidity—
Dissolve 500mg in 15mLof water,add 1drop of methyl red TS,and titrate with 0.020Nsodium hydroxide:not more than 0.50mLis required to produce a yellow color.
Water,Method Iá921ñ:
between 10.4%and 13.4%is found.
Residue on ignition á281ñ:
not more than 0.1%,from 500mg.
Chloride—
To 10mLof a solution (1in 100)add 1mLof 2Nnitric acid and 1mLof silver nitrate TS:no precipitate or turbidity is produced immediately.
Ammonium salts—
Heat 200mg with 5mLof 1Nsodium hydroxide on a steam bath for 1minute:no odor of ammonia is perceptible.
Limit of foreign alkaloids—
Dissolve 1.00g in 10mLof 1Nsodium hydroxide in a separator,and shake the solution with three successive portions of 15,10,and 10mLof chloroform,passing the chloroform solutions through a small filter previously moistened with chloroform.Shake the combined chloroform solutions with 5mLof water,separate the chloroform layer,and carefully evaporate on a steam bath to dryness.To the residue add 10.0mLof 0.020Nsulfuric acid,and heat gently until dissolved.Cool,add 2drops of methyl red TS,and titrate the excess acid with 0.020Nsodium hydroxide:not less than 7.5mLis required (1.5%).
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Assay—
Mobile phase—
Dissolve 0.73g of sodium 1-heptanesulfonate in 720mLof water,add 280mLof methanol and 10mLof glacial acetic acid,mix,filter,and degas.Make adjustments if necessary (seeSystem SuitabilityunderChromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Morphine Sulfate RSinMobile phase,and dilute quantitatively,and stepwise if necessary,withMobile phase to obtain a solution having a known concentration of about 0.24mg per mL.Prepare a fresh solution daily.
System suitability preparation—
Dissolve suitable quantities of USP Morphine Sulfate RSand phenol inMobile phase to obtain a solution containing about 0.24and 0.15mg per mL,respectively.
Assay preparation—
Transfer about 24mg of Morphine Sulfate,accurately weighed,to a 100-mLvolumetric flask,dissolve in and dilute withMobile phase to volume,and mix.
Chromatographic system(see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 284-nm detector and a 3.9-mm ×30-cm column that contains packing L1.The flow rate is about 1.5mLper minute.Chromatograph theStandard preparation and theSystem suitability preparation,and record the peak responses as directed forProcedure:the tailing factor for the morphine sulfate peak is not more than 2.0;the resolution,R,between the phenol and morphine sulfate peaks is not less than 2.0;and the relative standard deviation for replicate injections of theStandard preparation is not more than 2.0%.The relative retention times are about 0.7for phenol and 1.0for morphine sulfate.
Procedure—
Separately inject equal volumes (about 25µL)of theStandard preparation and theAssay preparation into the chromatograph,record the chromatograms,and measure the responses for the major peaks.Calculate the quantity,in mg,of (C17H19NO3)2·H2SO4in the portion of Morphine Sulfate taken by the formula:
100C(rU/rS),
in whichCis the concentration,in mg per mL,of anhydrous morphine sulfate in theStandard preparation,as determined from the concentration of USP Morphine Sulfate RScorrected for moisture content by a titrimetric water determination;andrUandrSare the peak responses obtained from theAssay preparation and theStandard preparation,respectively.
Auxiliary Information—
Staff Liaison:Clydewyn M.Anthony,Ph.D.,Scientist
Expert Committee:(PA2)Pharmaceutical Analysis 2
USP28–NF23Page 1314
Pharmacopeial Forum:Volume No.30(5)Page 1639
Phone Number:1-301-816-8139