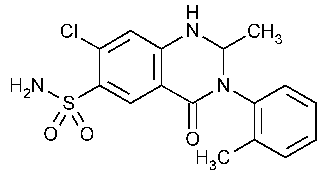
Metolazone
6-Quinazolinesulfonamide,7-chloro-1,2,3,4-tetrahydro-2-methy1-3-(2-methylphenyl)-4-oxo-.
7-Chloro-1,2,3,4-tetrahydro-2-methyl-4-oxo-3-o-tolyl-6-quinazolinesulfonamide [17560-51-9].
»Metolazone contains not less than 97.0percent and not more than 102.0percent of C16H16ClN3O3S,calculated on the dried basis.
Packaging and storage—
Preserve in tight,light-resistant containers.
Identification—
A:
Infrared Absorption á197Kñ.
Loss on drying á731ñ—
Dry it at 105 for 2hours:it loses not more than 1.0%of its weight.
for 2hours:it loses not more than 1.0%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Heavy metals,Method IIá231ñ:
0.0015%.
Chromatographic purity—
[NOTE—Protect Metolazone solutions from light.]
Standard preparations—
Dissolve an accurately weighed quantity of USP Metolazone RSin tetrahydrofuran and mix to obtain Standard preparation Ahaving a known concentration of 0.50mg per mL.Dilute a portion of Standard preparation Aquantitatively with tetrahydrofuran to obtain Standard preparation Bhaving a known concentration of 0.25mg per mL.
Test preparation—
Dissolve an accurately weighed quantity of Metolazone in tetrahydrofuran to obtain a solution containing 50mg per mL.
Procedure—
Separately apply 10µLof the Test preparationand each of the two Standard preparationsto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel mixture.Allow the spots to dry,and develop the chromatogram in a solvent system consisting of a mixture of chloroform,ethyl acetate,and formic acid (55:40:5)until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the developing chamber,mark the solvent front,air-dry,examine the plate under short-wavelength UVlight,and compare the intensities of any secondary spots observed in the chromatogram of the Test preparationwith those of the principal spots in the chromatograms of the Standard preparations:no secondary spot from the chromatogram of the Test preparationis larger or more intense than the principal spot obtained from Standard preparation B(0.5%)and the sum of the intensities of the secondary spots obtained from the Test preparationcorresponds to not more than 1.0%.
Assay—
[NOTE—Use low-actinic glassware throughout the Assay.]
Standard preparation—
Dissolve an accurately weighed quantity of USP Metolazone RSin methanol to obtain a solution having a known concentration of about 40µg per mL.
Assay preparation—
Transfer about 50mg of Metolazone,accurately weighed,to 100-mLvolumetric flask,dilute with methanol to volume,and mix.Pipet 20mLinto 250-mLvolumetric flask,dilute with methanol to volume,and mix.
Procedure—
Concomitantly determine the absorbances of the solutions at the wavelength of maximum absorbance at about 343nm,with a suitable spectrophotometer,using methanol as the blank.Calculate the quantity,in mg,of C16H16ClN3O3Sin the portion of Metolazone taken by the formula:
1.25C(AU/AS),
in which Cis the concentration,in µg per mL,of USP Metolazone RSin the Standard preparation;and AUand ASare the absorbances of the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Andrzej Wilk,Ph.D.,Senior Scientific Associate
Expert Committee:(PA5)Pharmaceutical Analysis 5
USP28–NF23Page 1277
Phone Number:1-301-816-8305