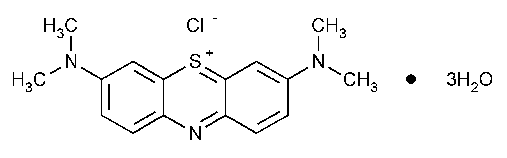
Methylene Blue
Phenothiazin-5-ium,3,7-bis(dimethylamino)-,chloride,trihydrate.
C.I.Basic Blue 9trihydrate [7220-79-3].
Anhydrous 319.86 [61-73-4].
»Methylene Blue contains not less than 98.0percent and not more than 103.0percent of C16H18ClN3S,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.Store at 25 ,excursions permitted between 15
,excursions permitted between 15 and 30
and 30 .
.
Identification,Infrared Absorption á197Kñ.
Loss on drying á731ñ—
Dry it at 75 and at a pressure not exceeding 5mm of mercury for 4hours:it loses between 8.0%and 18.0%of its weight.
and at a pressure not exceeding 5mm of mercury for 4hours:it loses between 8.0%and 18.0%of its weight.
Residue on ignition á281ñ:
not more than 1.2%.
Arsenic,Method Iá211ñ—
Prepare the Test Preparationby mixing 0.375g with 10mLof water in the arsine generator flask.Add 15mLof nitric acid and 5mLof perchloric acid,mix,and heat cautiously to the production of strong fumes of perchloric acid.Cool,wash down the sides of the flask with water,and again heat to strong fumes.Again cool,wash down the sides of the flask,and heat to fumes.Cool,dilute with water to 52mL,and add 3mLof hydrochloric acid:the resulting solution meets the requirements of the test,the addition of 20mLof 7Nsulfuric acid specified for Procedurebeing omitted.The limit is 8ppm.
Copper or zinc—
Ignite 1.0g in a porcelain crucible,using as low a temperature as practicable,until all of the carbon is oxidized.Cool the residue,add 15mLof 2Nnitric acid,and boil for 5minutes.Filter the cooled solution,and wash any residue with 10mLof water.To the combined filtrate and washing add an excess of 6Nammonium hydroxide,and filter the solution into a 50-mLvolumetric flask.Wash the precipitate with small portions of water,adding the washings to the filtrate,dilute the solution with water to volume,and mix.To 25mLof the solution add 10mLof hydrogen sulfide TS:no turbidity is produced within 5minutes(absence of zinc).Any dark color produced does not exceed that of a control prepared by boiling a quantity of cupric sulfate,equivalent to 200µg of copper,with 15mLof 2Nnitric acid for 5minutes and by treating this solution as directed above,beginning with “Filter the cooled solution”(0.02%of copper).
Chromatographic purity—
Quantitatively dissolve an accurately weighed quantity of Methylene Blue in methanol to obtain a Test solutioncontaining 1.0mg per mL.Dissolve a suitable quantity of USP Methylene Blue RSin methanol to obtain a Standard solutionhaving a concentration of 100µg per mL.Quantitatively dilute a portion of this solution with methanol to obtain a Diluted standard solutionhaving a concentration of 10µg per mL.Apply 5µLeach of the Test solution,the Standard solution,and the Diluted standard solutionto a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of octadecylsilanized chromatographic silica gel.Allow the spots to dry,and develop the chromatogram in a chromatographic chamber with a solvent system consisting of a mixture of the upper layer separated from a well-shaken mixture of water,n-butanol,and glacial acetic acid (100:80:20),until the solvent front has moved about three-fourths of the length of the plate.Remove the plate from the chamber,allow the solvent to evaporate,and visually locate the spots on the plate:the RFvalue of the principal spot in the chromatogram from the Test solutioncorresponds to that from the Standard solution,and other spots,if present in the chromatogram from the Test solution,consist of a secondary spot that does not exceed in size or intensity,the principal spot obtained from the Standard solution(10%),and not more than two additional spots,neither of which exceeds in size or intensity the principal spot from the Diluted standard solution(1%).
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Assay—
Transfer about 100mg of Methylene Blue,accurately weighed,to a 250-mLvolumetric flask,dissolve in and dilute with diluted alcohol to volume,and mix.Transfer 5.0mLof this solution to a 100-mLvolumetric flask,dilute with diluted alcohol to volume,and mix.Transfer 5.0mLof this solution to a 50-mLvolumetric flask,dilute with diluted alcohol to volume,and mix.This solution contains about 2µg per mL.Dissolve an accurately weighed quantity of USP Methylene Blue RSin diluted alcohol,and dilute quantitatively and stepwise with diluted alcohol to obtain a Standard solution having a known concentration of about 2µg per mL.Concomitantly determine the absorbances of both solutions in 1-cm cells at the wavelength of maximum absorbance at about 663nm,with a suitable spectrophotometer,using diluted alcohol as the blank.Calculate the quantity,in mg,of C16H18ClN3Sin the Methylene Blue taken by the formula:
50C(AU/AS),
in which Cis the concentration,in µg per mL,of anhydrous methylene blue in the Standard solution;and AUand ASare the absorbances of the solution of Methylene Blue and the Standard solution,respectively.
Auxiliary Information—
Staff Liaison:Andrzej Wilk,Ph.D.,Senior Scientific Associate
Expert Committee:(PA5)Pharmaceutical Analysis 5
USP28–NF23Page 1263
Pharmacopeial Forum:Volume No.29(5)Page 1534
Phone Number:1-301-816-8305