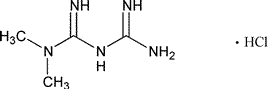
Add the following:
Imidodicarbonimidic diamide,N,N-dimethyl-,monohydrochloride.
1,1-Dimethylbiguanide monohydrochloride [1115-70-4].
»Metformin Hydrochloride contains not less than 98.5percent and not more than 101.0percent of C4H11N5·HCl,calculated on the dried basis.
Identification—
A:Infrared Absorption á197Kñ.
B:
It meets the requirements of the tests for Chloride á191ñ.
Loss on drying á731ñ—
Dry it at 105 for 5hours:it loses not more than 0.5%of its weight.
for 5hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Heavy metals,Method Iá231ñ:
0.001%.
Related compounds—
Mobile phase—
Prepare a solution in water,containing 17g of monobasic ammonium phosphate per L,adjust with phosphoric acid to a pHof 3.0,and mix.
Standard solution—
Prepare a solution of USP Metformin Related Compound A RSin water having a known concentration of about 0.2mg per mL.Transfer 1.0mLof this solution to a 200-mLvolumetric flask,dilute with Mobile phaseto volume,and mix.[NOTE—Metformin related compound Ais 1-cyanoguanidine.]
Test solution—
Transfer about 500mg of Metformin Hydrochloride,accurately weighed,to a 100-mLvolumetric flask,dissolve in and dilute with Mobile phase to volume,and mix.
Diluted test solution—
Transfer 1.0mLof the Test solutionto a 10-mLvolumetric flask,dilute with Mobile phaseto volume,and mix.Transfer 1.0mLof this solution to a 100-mLvolumetric flask,dilute with Mobile phaseto volume,and mix.
Resolution solution—
Transfer about 10mg of melamine to a 100-mLvolumetric flask,and dissolve in about 90mLof water.Add 5.0mLof theTest solution,dilute with water to volume,and mix.Transfer 1.0mLof this solution to a 50-mLvolumetric flask,dilute with Mobile phaseto volume,and mix.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 218-nm detector and a 4.6-mm ×25-cm column containing packing L9.The flow rate is about 1.0to 1.7mLper minute.Chromatograph the Resolution solution,and record the peak responses as directed for Procedure:the resolution,R,between melamine and metformin is not less than 10.
Procedure—
Separately inject equal volumes (about 20µL)of the Test solution,the Standard solution,and the Diluted test solutioninto the chromatograph,record the chromatograms for not less than twice the retention time of metformin,and measure the peak areas.The area of a peak corresponding to metformin related compound Ain the chromatogram of the Test solution is not greater than the area of the corresponding peak in the chromatogram of the Standard solution:not more than 0.02%of metformin related compound Ais found.
The area of any other secondary peak in the chromatogram of the Test solution is not greater than the area of the major peak in the chromatogram of the Diluted test solution;and the sum of the areas of all secondary peaks in the chromatogram of the Test solutionis not greater than five times the area of the major peak in the chromatogram of the Diluted test solution:not more than 0.1%of any other impurity is found;and not more than 0.5%of total impurities is found.
Assay—
[NOTE—To avoid overheating of the reaction medium,mix thoroughly throughout the titration,and stop the titration immediately after the endpoint has been reached.]Dissolve about 60mg of Metformin Hydrochloride,accurately weighed,in 4mLof anhydrous formic acid.Add 50mLof acetic anhydride.Titrate with 0.1Nperchloric acid VS,determining the endpoint potentiometrically.Perform a blank determination,and make any necessary correction (see Titrimetry á541ñ).Each mLof 0.1Nperchloric acid is equivalent to 8.28mg of C4H11N5·HCl. USP28
USP28
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(PA4)Pharmaceutical Analysis 4
USP28–NF23Page 1231
Pharmacopeial Forum:Volume No.29(6)Page 1925
Phone Number:1-301-816-8251