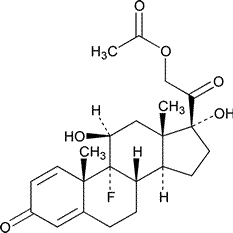
Isoflupredone Acetate
»Isoflupredone Acetate contains not less than 97.0percent and not more than 103.0percent of C23H29FO6,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed,light-resistant containers.
Labeling—
Label it to indicate that it is intended for veterinary use only.Where it is intended for use in preparing injectable dosage forms,the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
Identification,Infrared Absorption á197Mñ.
Absorptivity—
Test preparation:
25mg in 2000mLof alcohol.
Procedure—
Proceed as directed under Spectrophotometry and Light-Scattering á851ñ,and measure the absorbance at 240nm:the absorptivity is between 35.0and 38.0.
Specific rotation á781Sñ:
between +110 and +120
and +120 .
.
Test solution:
10mg per mL,in dioxane.
Bacterial endotoxins á85ñ—
Where the label states that Isoflupredone Acetate is sterile or that it must be subjected to further processing during the preparation of injectable dosage forms,it contains not more than 125USP Endotoxin Units per mg of isoflupredone acetate.
Loss on drying á731ñ—
Dry it at 105 for 4hours:it loses not more than 1.0%of its weight.
for 4hours:it loses not more than 1.0%of its weight.
Residue on ignition á281ñ:
not more than 0.5%.
Chromatographic purity—
Solution A—
Prepare a mixture of water,methanol,acetonitrile,and glacial acetic acid (500:350:150:3),and degas.
Solution B—
Prepare a mixture of acetonitrile,methanol,and water (550:500:3),and degas.
Mobile phase—
Use variable mixtures of Solution Aand Solution Bas directed for Chromatographic system.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
System suitability solution—
Dissolve accurately weighed quantities of USP Isoflupredone Acetate RSand USP Prednisolone Acetate RSin Solution Ato obtain a solution having known concentrations of about 0.03mg of each per mL.Sonicate,if necessary,to dissolve.
Test solution—
Dissolve an accurately weighed quantity of Isoflupredone Acetate in Solution Ato obtain a solution having a concentration of about 0.3mg per mL.Sonicate,if necessary,to dissolve.Use this solution within 16hours.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 254-nm detector and a 4.6-mm ×25-cm column that contains packing L7.The flow rate is about 1mLper minute.Protect the column from temperature fluctuations.The chromatograph is programmed as follows.
Chromatograph the System suitability solution,and record the peak areas as directed for Procedure:the retention time for isoflupredone acetate is between 21and 26minutes;the relative retention times are about 1.1for prednisolone acetate and 1.0for isoflupredone acetate;the resolution,R,between isoflupredone acetate and prednisolone acetate is not less than 1.2;and the column efficiency determined from isoflupredone is not less than 6000theoretical plates.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
| 0 | 100 | 0 | equilibration |
| 0–32.5 | 100 | 0 | isocratic |
| 32.5–47.5 | 100®0 | 0®100 | linear gradient |
| 47.5–50.5 | 0 | 100 | isocratic |
| 50.5–51.5 | 0®100 | 100®0 | linear gradient |
| 51.5–61.5 | 100 | 0 | isocratic |
Procedure—
Inject a volume (about 50µL)of the Test solutioninto the chromatograph,record the chromatogram,and measure the areas for the major peaks.Calculate the percentage of each impurity in the portion of Isoflupredone Acetate taken by the formula:
100(ri/rs),
in which riis the peak response for each impurity;and rsis the sum of the responses of all the peaks:not more than 1.0%of any individual impurity is found;and not more than 2.0%of total impurities is found,excluding those that are present in amounts less than 0.05%.
Other requirements—
Where the label states that it is sterile,it meets the requirements for Sterility Tests á71ñwhen tested as directed for Direct Transfer Methodunder Test Procedures.
Assay—
Mobile phase—
Prepare a mixture of n-butyl chloride,water-saturated n-butyl chloride,tetrahydrofuran,methanol,and glacial acetic acid (475:475:70:35:30).Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Diluent—
Use water-saturated chloroform.
Internal standard solution—
Dissolve an accurately weighed quantity of fluoxymesterone in Diluentto obtain a solution having a known concentration of about 0.9mg per mL.
Standard preparation—
Dissolve about 4mg of USP Isoflupredone Acetate RS,accurately weighed,in 8.0mLof Internal standard solutionand 32.0mLof Diluent.
Assay preparation—
Transfer about 4mg of Isoflupredone Acetate,accurately weighed,to a suitable container.Dissolve in 8.0mLof Internal standard solutionand 32.0mLof Diluent,centrifuge,and use the clear chloroform portion.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 254-nm detector and a 4-mm ×30-cm column that contains packing L3.The flow rate is about 0.7mLper minute.Chromatograph the Standard preparation,and record the peak areas as directed for Procedure:the relative retention times are about 1.0for isoflupredone acetate and 1.2for fluoxymesterone;the resolution,R,between isoflupredone acetate and fluoxymesterone is not less than 2.0;and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 12µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the quantity,in mg,of C23H29FO6in the portion of Isoflupredone Acetate taken by the formula:
WS(RU/RS),
in which WSis the weight,in mg,of USP Isoflupredone Acetate RStaken to prepare the Standard preparation;and RUand RSare the peak area ratios of isoflupredone acetate to fluoxymesterone obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Ian DeVeau,Ph.D.,Senior Scientist
Expert Committee:(VET)Veterinary Drugs
USP28–NF23Page 1068
Pharmacopeial Forum:Volume No.27(4)Page 2749
Phone Number:1-301-816-8178