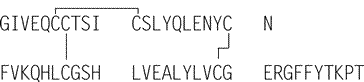Insulin Lispro
»Insulin Lispro is identical in structure to Insulin Human,except that it has lysine and proline at positions 28and 29,respectively,of chain B,whereas this sequence is reversed in Insulin Human.Insulin Lispro is produced by microbial synthesis via a recombinant DNAprocess.Its potency is not less than 27.0USP Insulin Lispro Units per mg,calculated on the dried basis.The proinsulin content of Insulin Lispro,determined by an appropriate and validated method,is not more than 10ppm.The host cell-derived protein content,determined by an appropriate and validated method,is not more than 10ppm.
NOTE—
One USP Insulin Lispro Unit is equivalent to 0.0347mg of pure Insulin Lispro.
Packaging and storage—
Preserve in tight containers,protected from light,and store in a freezer.
Labeling—
Label it to indicate that it has been prepared by microbial synthesis.
Identification—
A:
The retention time of the major peak in the chromatogram of the Assay preparationcorresponds to that in the chromatogram of the Standard preparation,as obtained in the Assay.
B:
Determine the peptide fragments,using the following peptide mapping procedure.
Sulfate buffer,HEPESbuffer,Mobile phase,Test digest solution,and Procedure—
Proceed as directed for Identificationtest Bunder Insulin Human.
Standard digest solution—
Proceed as directed for Identificationtest Bunder Insulin Human,except to use USP Insulin Lispro RSinstead of USP Insulin Human RS.
Chromatographic system—
Proceed as directed for Identificationtest Bunder Insulin Human,except to use the following elution program.
The flow rate is about 0.8mLper minute.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
| 0–3 | 95 | 5 | isocratic |
| 3–30 | 95®41 | 5®59 | linear gradient |
| 30–35 | 41®20 | 59®80 | linear gradient |
| 35–40 | 20®95 | 80®5 | return to initial |
| 40–50 | 95 | 5 | re-equilibration |
Bioidentity—
Proceed as directed for Bioidentity Testunder Insulin Assays á121ñ,except to obtain the first blood specimen at 45minutes,instead of 1hour,after the time of injection:meets the requirements.
Microbial limits á61ñ—
The total aerobic microbial count does not exceed 100per g,a portion of about 0.3g,accurately weighed,being used.
Bacterial endotoxins á85ñ:
not more than 10USP Endotoxin Units per mg,the kinetic-chromogenic method under Photometric Techniquesbeing used.
Loss on drying á731ñ—
Dry about 300mg,accurately weighed,at 105 for 16hours:it loses not more than 10.0%of its weight.
for 16hours:it loses not more than 10.0%of its weight.
Limit of high molecular weight proteins—
Proceed as directed in the test for Limit of high molecular weight proteinsunder Insulin:not more than 0.25%is found.
Related compounds—
Solvent—
Proceed as directed in the Assay.
Solution A—
Prepare a filtered and degassed mixture of Solventand acetonitrile (82:18).
Solution B—
Prepare a filtered and degassed mixture of Solventand acetonitrile (50:50).
Mobile phase—
Use variable mixtures of Solution Aand Solution Bas directed for Chromatographic system.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
System suitability solution—
Dissolve an accurately weighed quantity of Insulin Lispro in 0.01Nhydrochloric acid to obtain a solution containing about 3.5mg per mL.Allow to stand at room temperature to obtain a solution containing between 0.8%and 11%A-21desamido insulin lispro.
Test solution—
Dissolve about 3.5mg of Insulin Lispro in 1.0mLof 0.01Nhydrochloric acid.Store this solution for not more than 56hours in a refrigerator.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 214-nm detector and a 4.6-mm ×25-cm column that contains packing L1.The column temperature is maintained at 40 ,and the flow rate is about 1mLper minute.The chromatograph is programmed as follows.
,and the flow rate is about 1mLper minute.The chromatograph is programmed as follows.
Adjust theMobile phase composition and duration of the isocratic elution to obtain a retention time of about 41minutes for insulin lispro,with A-21desamido insulin lispro eluting near the start of the linear gradient phase.Chromatograph the System suitability solution,and record the peak responses as directed for Procedure:the resolution,R,between insulin lispro and A-21desamido insulin lispro is not less than 2.5;and the tailing factor for the insulin lispro peak is not more than 2.0.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
| 0–60 | 81 | 19 | isocratic |
| 60–83 | 81®51 | 19®49 | linear gradient |
| 83–84 | 51®81 | 49®19 | linear gradient |
| 84–94 | 81 | 19 | re-equilibration |
Procedure—
Proceed as directed for Procedurein the test for Related compoundsunder Insulin:not more than 1.00%of A-21desamido insulin lispro is found;not more than 0.50%of any other individual insulin lispro related compound is found;and not more than 2.00%of total impurities,excluding A-21desamido insulin lispro,is found.
Zinc content á591ñ—
Determine the zinc content of about 20mg of Insulin Lispro,accurately weighed:between 0.30%and 0.60%is found,calculated on the dried basis.
Assay—
Solvent—
Dissolve 28.4g of anhydrous sodium sulfate in 1000mLof water,mix,and adjust with phosphoric acid to a pHof 2.3.
Mobile phase—
Mix 745mLof Solventand 255mLof acetonitrile.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
System suitability solution—
Dissolve an accurately weighed quantity of Insulin Lispro in 0.01Nhydrochloric acid to obtain a solution having a concentration of about 1mg per mL.Allow to stand at room temperature to obtain a solution containing between 0.8%and 11%A-21desamido insulin lispro.
Standard preparation—
Dissolve an accurately weighed quantity of USP Insulin Lispro RSin 0.01Nhydrochloric acid to obtain a solution having a known concentration of about 0.7mg per mL.
Assay preparation—
Dissolve an accurately weighed portion of Insulin Lispro in 0.01Nhydrochloric acid to obtain a solution having a concentration of about 0.8mg per mL.
Chromatographic system (see Chromatography á621ñ)—
The liquid chromatograph is equipped with a 214-nm detector and a 4.6-mm ×10-cm column that contains packing L1.The column temperature is maintained at 40 ,and the flow rate is about 0.8mLper minute.Adjust the Mobile phaseto provide a retention time of about 24minutes for the main insulin lispro peak.Chromatograph three replicate injections of the System suitability solution,and record the peak responses as directed for Procedure:the resolution,R,between insulin lispro and A-21desamido insulin lispro is not less than 3.0;the tailing factor for the insulin lispro peak is not more than 1.5;and the relative standard deviation for replicate injections is not more than 1.1%.
,and the flow rate is about 0.8mLper minute.Adjust the Mobile phaseto provide a retention time of about 24minutes for the main insulin lispro peak.Chromatograph three replicate injections of the System suitability solution,and record the peak responses as directed for Procedure:the resolution,R,between insulin lispro and A-21desamido insulin lispro is not less than 3.0;the tailing factor for the insulin lispro peak is not more than 1.5;and the relative standard deviation for replicate injections is not more than 1.1%.
Procedure—
Separately inject equal volumes (about 20µL)of the Standard preparationand the Assay preparationinto the chromatograph,record the chromatograms,and measure the areas for the major peaks.Calculate the potency,in USP Insulin Lispro Units per mg,on the as-is basis by the formula:
(CS/CU)(rU/rS),
in which CSis the concentration,in USP Insulin Lispro Units per mL,of USP Insulin Lispro RSin the Standard preparation;CUis the concentration,in mg per mL,of Insulin Lispro in the Assay preparation;and rUand rSare the insulin lispro peak areas obtained from the Assay preparationand the Standard preparation,respectively.From the value obtained in the test for Loss on drying,calculate the potency on the dried basis.
Auxiliary Information—
Staff Liaison:Larry N.Callahan,Ph.D.,Scientist
Expert Committee:(BNT)Biotechnology and Natural Therapeutics/Diagnostics
USP28–NF23Page 1023
Pharmacopeial Forum:Volume No.28(4)Page 1125
Phone Number:1-301-816-8385