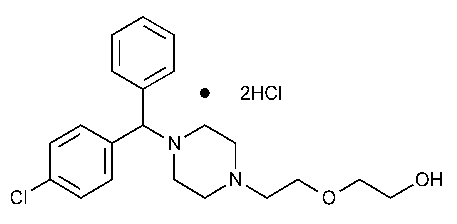
Hydroxyzine Hydrochloride
Ethanol,2-[2-[4-[(4-chlorophenyl)phenylmethyl]-1-piperazinyl]ethoxy]-,dihydrochloride,(±)-.
(±)-2-[2-[4-(p-Chloro-a-phenylbenzyl)-1-piperazinyl]ethoxy]ethanol dihydrochloride [2192-20-3].
»Hydroxyzine Hydrochloride,contains not less than 98.0percent and not more than 100.5percent of C21H27ClN2O2·2HCl,calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
USP Reference standards á11ñ—
USPp-Chlorobenzhydrylpiperazine RS.USP Hydroxyzine Hydrochloride RS.
Identification—
A:
Infrared Absorption á197Kñ.
B:
Ultraviolet Absorption á197Uñ—
Solution:
10µg per mL.
Medium:
alcohol.
Absorptivities at 230nm,calculated on the dried basis,do not differ by more than 3.0%.
C:
To 10mLof a solution (1in 400)add 2drops of nitric acid and 1mLof silver nitrate TS:a curdy,white precipitate,insoluble in 2Nnitric acid,but soluble in 6Nammonium hydroxide,separates (presence of chloride).
Loss on drying á731ñ—
Dry it in vacuum at 75 for 3hours:it loses not more than 5.0%of its weight.
for 3hours:it loses not more than 5.0%of its weight.
Residue on ignition á281ñ:
not more than 0.5%.
Heavy metals,Method IIá231ñ:
0.002%.
Chromatographic purity—
Mobile phase—
Prepare a filtered and degassed mixture of acetonitrile and 0.12Nsulfuric acid (90:10).Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Hydroxyzine Hydrochloride RSquantitatively in Mobile phaseto obtain a solution having a known concentration of about 1.8µg per mL.
Resolution solution—
Dissolve suitable quantities of USP Hydroxyzine Hydrochloride RSand USPp-Chlorobenzhydrylpiperazine RSin Mobile phaseto obtain a solution containing 3.6µg of each per mL.
Test preparation—
Transfer an accurately weighed quantity of Hydroxyzine Hydrochloride to a suitable volumetric flask,dissolve in and dilute with Mobile phaseto volume to obtain a solution containing a known concentration of about 0.6mg of specimen per mL,and mix.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 230-nm detector,and two series-coupled 3-mm ×10-cm columns that contain packing L3.The flow rate is about 0.4mLper minute.Chromatograph the Resolution solutionand the Standard preparation,and record the peak responses as directed under Procedure:the resolution,R,between the p-chlorobenzhydrylpiperazine and hydroxyzine peaks is not less than 1.2,and the relative standard deviation for replicate injections of the Standard preparationis not more than 2.0%.
Procedure—
Separately inject equal volumes (about 20µL)of the Standard preparationand the Test preparationinto the chromatograph,record the chromatograms for a total time of not less than 1.8times the retention time of the hydroxyzine peak,and measure the response for each peak,except for the main hydroxyzine peak in the chromatogram obtained from the Test preparation.Calculate the apparent percentage of each impurity in the specimen taken by the formula:
0.1(CS/CU)(rU/rS),
in which CSis the concentration,in µg per mL,of USP Hydroxyzine Hydrochloride RSin the Standard preparation;CUis the concentration,in mg per mL,of specimen in the Test preparation;rUis the peak response of a given impurity in the chromatogram obtained from the Test preparation;and rSis the peak response of hydroxyzine in the chromatogram obtained from the Standard preparation:not more than 0.3%of any impurity is found,and the sum of all impurities found is not greater than 1.5%.
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Assay—
Dissolve about 80mg of Hydroxyzine Hydrochloride,accurately weighed,in 50mLof a mixture of acetic anhydride and glacial acetic acid (7:3),and titrate with 0.1Nperchloric acid VS,determining the endpoint potentiometrically,using a glass electrode and a silver-silver chloride electrode containing saturated lithium perchlorate and saturated silver chloride in glacial acetic acid (see Titrimetry á541ñ).Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 22.39mg of C21H27ClN2O2·2HCl.
Auxiliary Information—
Staff Liaison:Salvador C.Salado,M.S.,Scientist and Latin American Liaison
Expert Committee:(PA3)Pharmaceutical Analysis 3
USP28–NF23Page 980
Pharmacopeial Forum:Volume No.29(6)Page 1902
Phone Number:1-301-816-8165