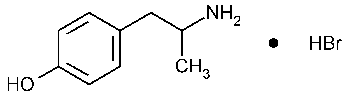
Hydroxyamphetamine Hydrobromide
Phenol,4-(2-aminopropyl)-,hydrobromide.
(±)-p-(2-Aminopropyl)phenol hydrobromide [306-21-8].
»Hydroxyamphetamine Hydrobromide contains not less than 98.0percent and not more than 101.5percent of C9H13NO·HBr,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed,light-resistant containers.
Identification—
A:
Infrared Absorption á197Kñ.
B:
Dissolve about 500mg of ammonium molybdate in 10mLof sulfuric acid,and add to this solution about 2mg of Hydroxyamphetamine Hydrobromide:an intense blue color is produced (distinction from similar amino compounds such as amphetamine and methamphetamine,which,lacking a phenolic hydroxyl,do not undergo this reaction).
C:
Dissolve about 200mg in 2mLof water,and add a solution of 500mg of potassium carbonate in 2mLof water.Extract with two 10-mLportions of ether,allow the clear ether solution to evaporate to dryness,and dry at about 80 :the hydroxyamphetamine so obtained melts between 124
:the hydroxyamphetamine so obtained melts between 124 and 127
and 127 (see Class Iunder Melting Range or Temperature á741ñ).
(see Class Iunder Melting Range or Temperature á741ñ).
D:
To a solution of about 10mg of it in 10mLof water add 1mLof 2Nnitric acid,then add silver nitrate TS:a pale yellow precipitate is formed,and it is slightly soluble in 6Nammonium hydroxide.
Loss on drying á731ñ—
Dry it at 105 for 2hours:it loses not more than 0.5%of its weight.
for 2hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Bromide content—
Accurately weigh about 400mg,and dissolve in 50mLof water.Add 50mLof methanol and 10mLof glacial acetic acid,then add eosin Y TS,and titrate with 0.1Nsilver nitrate VS.Each mLof 0.1Nsilver nitrate is equivalent to 7.990mg of Br:the content of Br,calculated on the dried basis,is between 33.6%and 35.2%.
Ordinary impurities á466ñ—
Test solution:
methanol.
Standard solution:
methanol.
Eluant:
a mixture of toluene,methanol,and ammonium hydroxide (10:4:0.25).
Visualization:
1.
Assay—
Dissolve about 400mg of Hydroxyamphetamine Hydrobromide,accurately weighed,in a mixture of 10mLof glacial acetic acid and 10mLof mercuric acetate TS,warming slightly,if necessary,to effect solution.Add crystal violet TS,and titrate with 0.1Nperchloric acid VS.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 23.21mg of C9H13NO·HBr.
Auxiliary Information—
Staff Liaison:Lawrence Evans,III,Ph.D.,Scientist
Expert Committee:(PA6)Pharmaceutical Analysis 6
USP28–NF23Page 976
Phone Number:1-301-816-8389