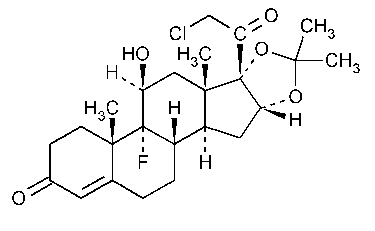
Halcinonide
Pregn-4-ene-3,20-dione,21-chloro-9-fluoro-11-hydroxy-16,17-[(1-methylethylidene)bis(oxy)]-,(11b,16a)-.
21-Chloro-9-fluoro-11b,16a,17-trihydroxypregn-4-ene-3,20-dione cyclic 16,17-acetal with acetone [3093-35-4].
»Halcinonide contains not less than 97.0percent and not more than 102.0percent of C24H32ClFO5.
Packaging and storage—
Preserve in well-closed containers.
Identification,Infrared Absorption á197Kñ.
Specific rotation á781Sñ:
between +150 and +160
and +160 .
.
Test solution:
20mg per mL,in chloroform.
Loss on drying á731ñ—
Dry it in vacuum at 100 for 3hours:it loses not more than 1.0%of its weight.
for 3hours:it loses not more than 1.0%of its weight.
Residue on ignition á281ñ:
not more than 0.2%.
Chromatographic purity—
Prepare the test solution by dissolving 50mg of Halcinonide in 5.0mLof a mixture of chloroform and methanol (1:1).Divide the area of a suitable thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel mixture,into three equal sections,the first two sections to be used for the test solution and the third section for the blank.Apply 100µLof the test solution to appropriate sections of the plate,drying each solution as it is applied with a current of warm air.Using a continuous elution chromatographic chamber,develop the chromatogram in a solvent system consisting of a mixture of chloroform and ethyl acetate (5:1)for about 2hours.Remove the plate from the developing chamber,dry in an oven at 90 for 15minutes,and locate the bands by viewing under short-wavelength UVlight.Mark the principal band and any secondary bands.Quantitatively remove the silica gel containing these bands,including a corresponding blank segment,and transfer to separate glass-stoppered,50-mLcentrifuge tubes,combining the impurities if more than one impurity is present.Add 30.0mLof dehydrated alcohol to the tubes containing the principal band and the corresponding blank,and add 10.0mLof dehydrated alcohol to the tubes containing the combined impurities and the corresponding blank.Insert stoppers in the tubes,and shake gently on a reciprocating shaker for about 60minutes.Centrifuge,dilute the principal band eluate and its corresponding blank eluate with an equal volume of dehydrated alcohol,and mix.Determine the absorbances of the clear supernatant eluates in 1-cm cells at the wavelength of maximum absorbance at about 239nm,with a suitable spectrophotometer,using dehydrated alcohol as the blank.Calculate the percentage of chromatographic impurities by the formula:
for 15minutes,and locate the bands by viewing under short-wavelength UVlight.Mark the principal band and any secondary bands.Quantitatively remove the silica gel containing these bands,including a corresponding blank segment,and transfer to separate glass-stoppered,50-mLcentrifuge tubes,combining the impurities if more than one impurity is present.Add 30.0mLof dehydrated alcohol to the tubes containing the principal band and the corresponding blank,and add 10.0mLof dehydrated alcohol to the tubes containing the combined impurities and the corresponding blank.Insert stoppers in the tubes,and shake gently on a reciprocating shaker for about 60minutes.Centrifuge,dilute the principal band eluate and its corresponding blank eluate with an equal volume of dehydrated alcohol,and mix.Determine the absorbances of the clear supernatant eluates in 1-cm cells at the wavelength of maximum absorbance at about 239nm,with a suitable spectrophotometer,using dehydrated alcohol as the blank.Calculate the percentage of chromatographic impurities by the formula:
100Ai/(Ai+6Au),
in which Aiis the absorbance of the combined impurity bands eluate,corrected for the corresponding blank,and Auis the absorbance of the principal band eluate,corrected for the corresponding blank.Not more than 3.0%is found.
Assay—
Standard preparation—
Dissolve an accurately weighed quantity of USP Halcinonide RSin methanol,and dilute quantitatively and stepwise with methanol to obtain a solution having a known concentration of about 15µg per mL.
Assay preparation—
Weigh accurately about 30mg of Halcinonide,transfer to a 100-mLvolumetric flask,dissolve in methanol,dilute with methanol to volume,and mix.Transfer 5.0mLof this solution to a second 100-mLvolumetric flask,dilute with methanol to volume,and mix.
Procedure—
Concomitantly determine the absorbances of the Assay preparationand the Standard preparationin 1-cm cells at the wavelength of maximum absorbance at about 239nm,with a suitable spectrophotometer,using methanol as the blank.Calculate the quantity,in mg,of C24H32ClFO5in the portion of Halcinonide taken by the formula:
2C(AU/AS),
in which Cis the concentration,in µg per mL,of USP Halcinonide RSin the Standard preparation,and AUand ASare the absorbances of the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:Clydewyn M.Anthony,Ph.D.,Scientist
Expert Committee:(PA2)Pharmaceutical Analysis 2
USP28–NF23Page 935
Phone Number:1-301-816-8139