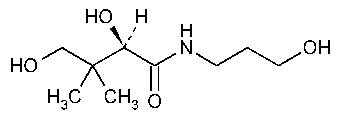
Dexpanthenol
Butanamide,2,4-dihydroxy-N-(3-hydroxypropyl)-3,3-dimethyl-,(R)-.
D-(+)-2,4-Dihydroxy-N-(3-hydroxypropyl)-3,3-dimethylbutyramide [81-13-0].
»Dexpanthenol contains not less than 98.0percent and not more than 102.0percent of C9H19NO4,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
Identification—
A:
Infrared Absorption á197Fñ.
B:
To 1mLof a 1in 10solution add 5mLof 1Nsodium hydroxide and 1drop of cupric sulfate TS,and shake vigorously:a deep blue color develops.
C:
To 1mLof a solution (1in 100)add 1mLof 1Nhydrochloric acid,and heat on a steam bath for about 30minutes.Cool,add 100mg of hydroxylamine hydrochloride,mix,and add 5mLof 1Nsodium hydroxide.Allow to stand for 5minutes,then adjust with 1Nhydrochloric acid to a pHof between 2.5and 3.0,and add 1drop of ferric chloride TS:a purplish red color develops.
Specific rotation á781Sñ:
between +29.0 and +31.5
and +31.5 .
.
Test solution:
50mg per mL,in water.
Refractive index á831ñ:
between 1.495and 1.502,at 20 .
.
Water,Method Iá921ñ:
not more than 1.0%.
Residue on ignition á281ñ:
not more than 0.1%.
Limit of aminopropanol—
Transfer about 5g,accurately weighed,to a 50-mLflask,and dissolve in 10mLof water.Add bromothymol blue TS,and titrate with 0.1Nsulfuric acid VSto a yellow endpoint.Each mLof 0.1Nsulfuric acid is equivalent to 7.5mg of aminopropanol.Not more than 1.0%is found.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Potassium biphthalate solution—
Dissolve 20.42g of potassium biphthalate in glacial acetic acid contained in a 1000-mLvolumetric flask.If necessary,warm the mixture on a steam bath to achieve complete solution,observing precautions against absorption of moisture.Cool to room temperature,dilute with glacial acetic acid to volume,and mix.
Procedure—
Transfer about 400mg of Dexpanthenol,accurately weighed,to a 300-mLflask fitted to a reflux condenser by means of a standard-taper glass joint,add 50.0mLof 0.1Nperchloric acid VS,and reflux for 5hours.Cool,observing precautions to prevent atmospheric moisture from entering the condenser,and rinse the condenser with glacial acetic acid,collecting the rinsings in the flask.Add 5drops of crystal violet TS,and titrate with Potassium biphthalate solutionto a blue-green endpoint.Perform a blank determination,and note the differences in volumes required.Each mLof the difference in volumes of 0.1Nperchloric acid consumed is equivalent to 20.53mg of C9H19NO4.
Auxiliary Information—
Staff Liaison:Lawrence Evans,III,Ph.D.,Scientist
Expert Committee:(DSN)Dietary Supplements:Non-Botanicals
USP28–NF23Page 600
Phone Number:1-301-816-8389