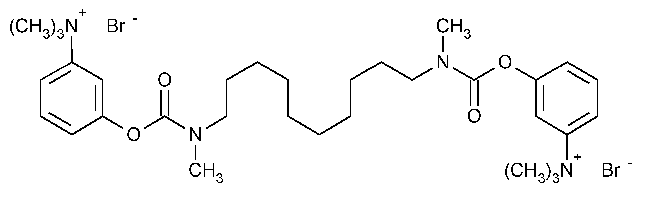
Demecarium Bromide
Benzenaminium,3,3¢-[1,10-decanediylbis[(methylimino)carbonyloxy]]bis[N,N,N-trimethyl]-,dibromide.
(m-Hydroxyphenyl)trimethylammonium bromide decamethylenebis[methylcarbamate](2:1) [56-94-0].
»Demecarium Bromide contains not less than 95.0percent and not more than 100.5percent of C32H52Br2N4O4,calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight,light-resistant containers.
Identification—
A:
Infrared Absorption á197Kñ.
B:
Dissolve about 100mg in 50mLof 1Nsodium hydroxide,and reflux for 15minutes.Cool,and add 3mLof the refluxed solution to 25mLof saturated sodium bicarbonate solution.Add,with mixing,4mLof N,N-dimethyl-p-phenylenediamine dihydrochloride solution (1.5in 10,000)and 2mLof sodium hypochlorite solution (1.5in 20,000):a violet-blue color is produced.
C:
Dissolve about 50mg in 20mLof water,add 10mLof a 1in 50solution of ammonium reineckate in methanol,and allow to stand for 30minutes with occasional swirling:a pink reineckate of demecarium forms,and it melts between 131 and 136
and 136 ,with decomposition.
,with decomposition.
D:
Asolution of it responds to the tests for Bromide á191ñ.
pHá791ñ:
between 5.0and 7.0,in a solution (1in 100).
Water,Method Iá921ñ:
not more than 2.0%.
Residue on ignition á281ñ:
not more than 0.1%.
Heavy metals,Method Iá231ñ:
0.002%.
Limit of m-trimethylammoniophenol bromide—
Control solution—
Dissolve 100mg of m-dimethylaminophenol in 10mLof alcohol in a 1000-mLvolumetric flask,dilute with water to volume,and mix.Pipet 1mLof this solution into a 500-mLvolumetric flask,dilute with water to volume,and mix.
Test solution—
Transfer 100mg of Demecarium Bromide to a 100-mLvolumetric flask,add water to volume,and mix.
Procedure—
Pipet 25mLof the Test solutioninto a glass-stoppered,50-mLcentrifuge tube,and pipet 25mLof the Control solutioninto a second,similar tube.To each tube add 3mLof pH7.0phosphate buffer (see under Solutionsin the section Reagents,Indicators,and Solutions),1mLof N,N-dimethyl-p-phenylenediamine dihydrochloride solution (1.5in 10,000),5mLof isobutyl alcohol,and 1mLof sodium hypochlorite solution (1.5in 20,000).Insert the stoppers in the tubes,shake the mixtures for 5minutes,and centrifuge:any blue color produced in the upper layer obtained from the Test solutionis not more intense than that obtained from the Control solution.
Assay—
Dissolve about 0.8g of Demecarium Bromide,accurately weighed,in a mixture of 75mLof glacial acetic acid and 15mLof mercuric acetate TS,warming slightly,if necessary,to effect solution.Add 2drops of crystal violet TS,and titrate with 0.1Nperchloric acid VS.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 35.83mg of C32H52Br2N4O4.
Auxiliary Information—
Staff Liaison:Ravi Ravichandran,Ph.D.,Senior Scientist
Expert Committee:(PA3)Pharmaceutical Analysis 3
USP28–NF23Page 576
Phone Number:1-301-816-8330