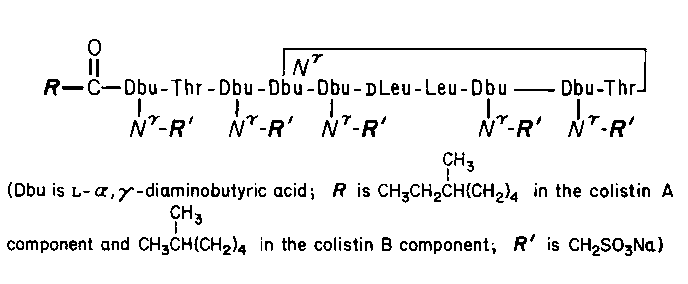Colistimethate Sodium
C57H103N16Na5O28S5(colistin Bcomponent)
1735.80
Colistimethate sodium.
Pentasodium colistinmethanesulfonate [8068-28-8;21362-08-3].
»Colistimethate Sodium has a potency equivalent to not less than 390µg of colistin per mg.
Labeling—
Where it is intended for use in preparing injectable dosage forms,the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
Constituted solution—
At the time of use,it meets the requirements for Constituted Solutionsunder Injections á1ñ.
Identification,Infrared Absorption á197Kñ.
pHá791ñ:
between 6.5and 8.5,in a solution containing 10mg per mL.
Loss on drying á731ñ—
Dry about 100mg,accurately weighed,in a capillary-stoppered bottle in vacuum at a pressure not exceeding 5mm of mercury at 60 for 3hours:it loses not more than 7.0%of its weight.
for 3hours:it loses not more than 7.0%of its weight.
Heavy metals,Method IIá231ñ:
not more than 0.003%.
Free colistin—
Dissolve 80mg in 3mLof water,and add 0.05mLof silicotungstic acid solution (1in 10):no immediate precipitate is formed.
Other requirements—
Where the label states that Colistimethate Sodium is sterile,it meets the requirements for Sterilityand Bacterial endotoxinsunder Colistimethate for Injection.Where the label states that Colistimethate Sodium must be subjected to further processing during the preparation of injectable dosage forms,it meets the requirements for Bacterial endotoxinsunder Colistimethate for Injection.
Assay—
Assay preparation—
Dissolve a suitable quantity of Colistimethate Sodium,accurately weighed,in 2.0mLof water,add a sufficient accurately measured volume of Buffer No.6to obtain a solution having a convenient concentration.
Procedure—
Proceed as directed for Colistimethate Sodium under Antibiotics—Microbial Assays á81ñ,using an accurately measured volume of Assay preparationdiluted quantitatively with Buffer No.6to yield a Test Dilutionhaving a concentration assumed to be equal to the median dose level of the Standard.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 540
Phone Number:1-301-816-8335